Abstract
-
Purpose
Transcatheter arterial embolization (TAE) is widely used for managing renal angiomyolipoma (AML) to prevent hemorrhage and control symptoms while preserving renal function. However, the optimal embolic material remains undetermined due to limited comparative data. This study aimed to compare the effectiveness of ethanol-based embolization versus polyvinyl alcohol (PVA) and to evaluate additional benefits of microcoil use in ethanol-based treatments.
-
Materials and Methods
We retrospectively analyzed 119 patients with single renal AML who underwent TAE at two tertiary centers between 2005 and 2023. Patients were grouped into ethanol-based (ethanol alone or ethanol plus microcoil, n = 93) and PVA (n = 26) cohorts. Subgroup analysis compared ethanol alone (n = 24) versus ethanol plus microcoil (n = 69). Inverse probability treatment weighting and linear mixed-effects models were used to assess tumor volume reduction and treatment response (≥50% volume reduction).
-
Results
After adjustment, the ethanol group demonstrated significantly greater tumor volume reduction than the PVA group at 6 and 12 months (adjusted mean difference = –23.9%, p = 0.002; –23.1%, p = 0.001) and a higher response rate (92.1% vs. 78.4%, p = 0.043). In the subgroup analysis, ethanol plus microcoil achieved higher response (91.3% vs. 73.8%; odds ratio [OR], 3.73; p = 0.038) and lower recurrence (7.1% vs. 30.2%; OR, 0.18; p = 0.008) compared with ethanol alone.
-
Conclusion
Ethanol-based embolization provides superior tumor control compared to PVA in renal AML, and the addition of microcoils enhances early volume reduction and reduces recurrence, supporting its use as an effective treatment strategy.
-
Keywords: Angiomyolipoma; Therapeutic embolization; Ethanol; Polyvinyl alcohol; Microcoil
Introduction
Renal angiomyolipoma (AML) is a benign mesenchymal tumor characterized by a composite structure of dysmorphic blood vessels, smooth muscle cells, and mature adipose tissue [
1-
3]. These tumors can manifest sporadically or in association with tuberous sclerosis complex, displaying a higher prevalence in women in sporadic cases [
2-
4]. Although AML typically shows subtle clinical manifestations, large tumors exceeding 4 cm or containing aneurysms larger than 5 mm pose a risk for severe complications such as rupture, hemorrhage, or renal failure [
4].
Transcatheter arterial embolization (TAE) is currently the favored nephron-sparing approach for AML treatment, utilized both prophylactically and emergently, particularly in high-risk bleeding situations [
2,
5-
7]. Various embolic agents have been employed, including particulate materials such as polyvinyl alcohol (PVA), liquid embolics (ethanol), and coils, aiming to reduce tumor vascularity and size while preserving renal function. Previous studies using liquid or particulate embolic agents reported various tumor reduction percentages, ranging from 55% to 77% [
8-
10]. However, no embolic agent has consistently demonstrated superior clinical outcomes, and comparative studies focusing on embolization materials remain scarce [
11-
15].
Therefore, we aimed to compare clinical outcomes of TAE using ethanol-based embolization (ethanol alone or ethanol plus microcoil) versus PVA particles in patients with single renal AML.
Materials and Methods
Patient
This study was approved by the Institutional Review Board (B-2412-944-104), and the need for consent was waived. From 2005 to 2023, electronic medical records of two tertiary academic hospitals (Seoul National University Bundang Hospital, Seongnam, Korea, and Jeonbuk University Hospital, Jeonju, Korea) were reviewed for patients who underwent TAE for renal AML. Inclusion criteria were adult (>18 years old), a single renal AML with maximal diameter ≥4 cm or symptomatic lesion with any size, preprocedural computed tomography (CT)/magnetic resonance imaging (MRI) within 3 months before TAE. Exclusion criteria were patients with ruptured AML before TAE, previous history of TAE for the lesion, multiple/bilateral lesions, contrast allergy or impaired renal function (estimated glomerular filtration rate < 30 mL/min/1.73 m2), lost to follow-up within 1 month after TAE. Baseline characteristics of enrolled patients, including age, sex, presence of a symptom, the date of preprocedural and follow-up cross-sectional image studies including CT or MRI, TAE-procedure date, and any occurrence or change of symptom after an embolization, were identified.
Transarterial Embolization
During the study period, seven interventional radiologists performed TAE for renal AML, and embolic materials were chosen based on the operator's preference. The procedure began with 5-Fr sheath inserted via right common femoral artery access under local anesthesia, followed by renal arteriography to evaluate tumor location and feeding arteries. A 2.0-Fr microcatheter (Progreat alpha, Terumo, Tokyo, Japan) was then advanced into the feeding artery, ensuring the preservation of normal renal parenchyma. In the PVA group, embolization was performed with 355–500 μm PVA particles (Contour, Boston Scientific, Marlborough, MA, USA). In the ethanol group, 99% absolute ethanol (Daihan Pharma, Seoul, Korea) was infused to ablate the vascular endothelium of the feeding arteries. In both groups, the embolization endpoint was the cessation of inflow to the target feeding arteries. In ethanol with microcoil group, 99% absolute ethanol embolization was first performed in a manner similar to the ethanol group. After confirming occlusion, one or two microcoils (2–3 mm; Concerto, Medtronic, Minneapolis, MN, USA; Tornado, Cook Medical, Bloomington, IN, USA) were placed in the proximal portion of the target vessels to prevent recanalization and achieve durable embolization. Final angiography confirmed the devascularization of the tumors in all groups.
Follow-up and Image Analysis
Follow-up was performed at 6 and 12 months after embolization and then at 1-year intervals thereafter, with CT or MRI. Two radiologists with more than 10 years of clinical experience reviewed the patients' preprocedural and follow-up CT/MRI images in consensus. The date of the cross-sectional imaging, the long and short diameters on the axial image, and the z-axis diameter on the coronal image of the target lesion were acquired. Tumor volume at each time point was calculated using the ellipse volume formula (long diameter × short diameter × z-axis diameter × 0.52). Tumor volume reduction was assessed using pre- and post-TAE imaging. A positive treatment response was defined as a volume reduction of the tumor by 50% or more after TAE [
16].
Technical success was defined as the successful cannulation of all feeding arteries and complete embolization of the targeted vascular network. Tumor volume changes were evaluated at each follow-up to assess therapeutic efficacy, and long-term efficacy was determined by tumor volume reduction at the 24-month follow-up. Multivariable logistic regression analysis was performed to identify independent predictors of achieving this response. Tumor recurrence was defined as an increase in tumor volume compared to the preceding follow-up, assessing the durability of embolization effects over time [
7].
Continuous variables were summarized using means ± standard deviation and categorical variables as frequencies and percentages. Differences between groups were compared with Student's t-test or Wilcoxon rank-sum test for continuous data, and chi-square or Fisher's exact test for categorical variables.
Missing follow-up data were addressed by multiple imputation using chained equations. Propensity scores were calculated using multivariable logistic regression incorporating age, sex, and baseline tumor volume to balance baseline differences. Inverse probability of treatment weighting (IPTW) based on these propensity scores was performed to adjust for confounding factors in the main comparison between ethanol and PVA groups. After IPTW, standardized mean differences <0.1 were considered indicative of adequate balance.
Linear mixed-effects modeling was used to assess longitudinal tumor volume changes over the follow-up periods. The models included random intercepts for subjects and fixed effects for treatment group, follow-up time, and their interactions. Additionally, within the ethanol group, subgroup analyses comparing Ethanol alone and Ethanol plus microcoil were conducted following the same statistical methodology.
Multivariable logistic regression analysis identified independent predictors of achieving treatment response, defined as a tumor volume reduction greater than 50%. A p-value <0.05 was considered statistically significant. All analyses were performed using SPSS version 21.0 (IBM Corp., Armonk, NY, USA) and R version 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient Demographics
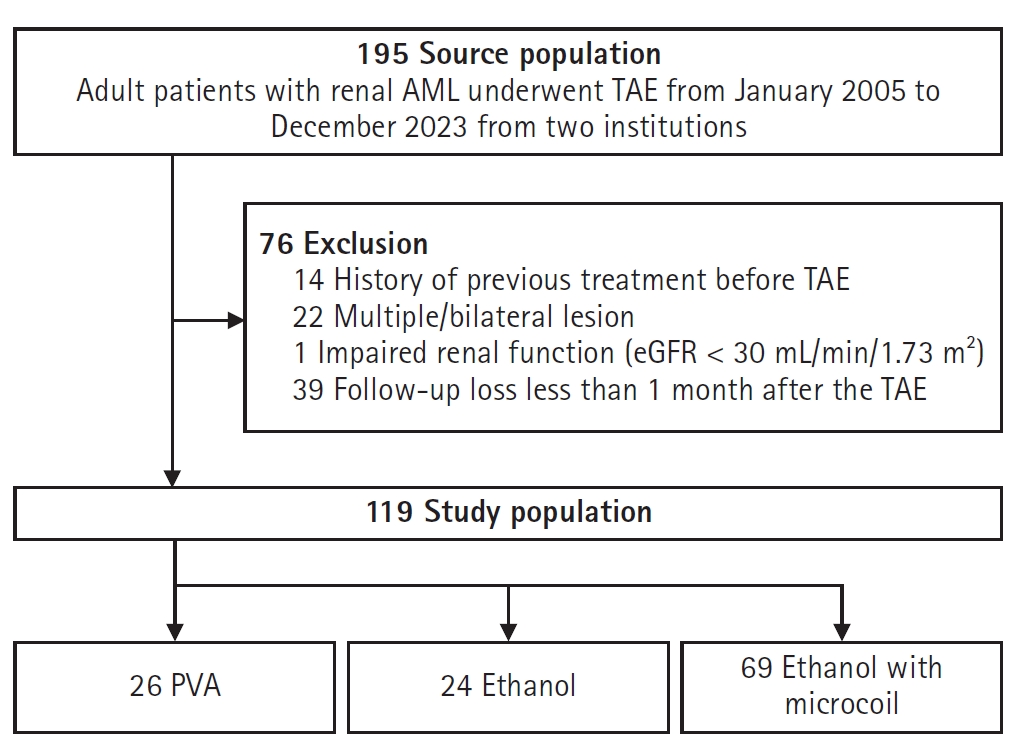
A total of 119 patients (mean age, 48.6 ± 13.3 years; 70.6% female) were enrolled (
Fig. 1). The mean follow-up period was 33.1 ± 26.5 months (median, 27.8 months), with group-specific means of 43.6 ± 37.2 months (PVA), 22.9 ± 14.2 months (ethanol), and 32.6 ± 23.4 months (ethanol with microcoil). Patients were categorized into two groups based on embolization agents: the ethanol group (n = 93, including ethanol alone (n = 24) and ethanol plus microcoil (n = 69) and the PVA group (n = 26). No significant differences in age or sex were observed between groups. However, baseline tumor volume tended to be larger in the PVA group compared to the ethanol group (262.8 ± 334.2 cm³ vs. 118.1 ± 158.6 cm³, p = 0.098). After IPTW adjustment, baseline characteristics including age, sex, and tumor volume were balanced (
Table 1).
Technical success was achieved in all cases, with complete embolization confirmed by final angiography. The embolization endpoints—cessation of inflow to target feeding arteries in the PVA and ethanol groups—were consistently reached in all groups. No immediate post-procedural complications were recorded.
Treatment Response and Recurrence
After IPTW adjustment, ethanol-based embolization achieved significantly greater tumor volume reduction compared with PVA embolization at 6 and 12 months (adjusted mean difference = –23.9%, p = 0.002; –23.1%, p = 0.001, respectively), whereas the difference was not significant at 24 months (p = 0.266). The ≥50% response rate was higher in the ethanol group than in the PVA group (92.1% vs. 78.4%; odds ratio [OR], 2.86; p = 0.043), with no difference in recurrence (p = 0.844) (
Table 2). In the subgroup analysis, ethanol + microcoil achieved a higher response rate (91.3% vs. 73.8%; OR, 3.73; p = 0.038) and lower recurrence (7.1% vs. 30.2%; OR, 0.18; p = 0.008) compared with ethanol alone, indicating improved treatment durability without significant differences in overall volume reduction (
Table 3).
Table 4 highlights the results of univariable and multivariable logistic regression analyses. The type of embolic material was an independent predictor of favorable treatment response. Patients treated with the ethanol with microcoil approach were significantly more likely to achieve ≥50% tumor volume reduction (adjusted odds ratio, 4.667; p = 0.011).
Discussion
This study demonstrated that embolization using ethanol with microcoil outperformed single-agent embolization with either PVA or ethanol in terms of treatment response and recurrence rate. Technical success was achieved in all cases, and the ethanol with microcoil group exhibited the highest treatment response rate (91.3%) and tumor volume reduction. These findings highlight the efficacy and durability of this ethanol with microcoil approach for managing renal AML.
Historically, embolization with PVA is one of the earliest embolic agents, with modest therapeutic effects and relatively higher recurrence rates attributed to incomplete vessel occlusion [
17,
18]. In contrast, ethanol-based embolization has yielded more favorable outcomes [
10,
12,
17,
18]. A recent meta-analysis of 13 studies (478 patients) reported that ethanol embolization produced the greatest tumor shrinkage (95.8%) and the lowest re-intervention rate (3.4%) [
18]. Theoretically, ethanol offers distinct advantages over particulate agents: as a liquid, it penetrates the distal microvasculature, produces endothelial destruction and tumor necrosis, and provides permanent occlusion at the arteriolar and capillary level.
However, ethanol-only embolization may result in incomplete occlusion due to rapid washout in high-flow vessels, increasing the risk of recanalization and recurrence [
13,
15]. The low viscosity of ethanol makes it difficult to control high-flow vessels, potentially compromising durable occlusion and necessitating adjunctive devices such as micro-balloons or coils.
In our study, these issues were addressed by combining ethanol with microcoil. Our study demonstrated superior outcomes with this combined ethanol and microcoil approach, achieving ≥50% tumor volume reduction in 91.3% of cases and the lowest recurrence rate of 7.1%, compared to ethanol-only or PVA-only treatments.
The enhanced efficacy of the ethanol with microcoil method likely stems from its mechanism of action. Ethanol initiates endothelial injury and thrombus formation, while the microcoil acts as a physical barrier to stabilize the thrombus and prevent washout, allowing for better contact time between ethanol and endothelium [
19]. This synergy ensures complete and durable vessel occlusion, a key factor in its superior outcomes. The significantly greater tumor volume reduction observed in the ethanol with microcoil group, even before IPTW adjustment, supports this mechanism. These findings are consistent with prior observations of embolization approaches that emphasize the importance of stable vessel closure [
3,
12].
Treatment response analysis revealed that the ethanol with microcoil strategy achieved 91.3% favorable outcomes, surpassing those of the PVA (69.2%) and ethanol (75%) groups. This is a promising result compared to the treatment response rates reported in earlier studies, which ranged from 60% to 80% with single-agent embolization [
12,
15,
17]. These findings highlight the potential of the ethanol with microcoil approach, offering enhanced efficacy and reduced recurrence risks.
The ethanol with microcoil group demonstrated the lowest recurrence rate, likely due to the combined effect of ethanol’s distal embolization, inducing endothelial injury and thrombosis, and microcoils’ proximal embolization, ensuring stable vascular occlusion. This dual strategy minimizes the risk of recanalization, offering superior long-term efficacy compared to single-modality approaches.
Multivariable analysis further reinforces the robustness of our results. After adjusting for confounding variables, the type of embolic material emerged as an independent predictor of favorable treatment response (≥50% tumor volume reduction). Specifically, patients treated with the ethanol with microcoil approach demonstrated significantly higher odds of achieving a positive treatment response than those in the PVA or ethanol groups.
Despite these results, this study's limitations should be acknowledged. As a retrospective analysis, inherent biases such as operator variability and loss to follow-up may affect the outcomes. However, this real-world setting reflects diverse operator expertise, enhancing the generalizability of the findings. Furthermore, variations in baseline tumor volume across groups were adjusted using IPTW, mitigating confounding effects. The study lacked long-term follow-up, leaving questions about the durability of outcomes unaddressed. Additionally, this study did not evaluate variations in tumor composition or preprocedural CT and angiographic findings—such as hypervascularity, aneurysm formation, or arteriovenous shunts—that may influence embolization strategy and outcomes. Minor complications, including post-embolization syndrome, were also not systematically recorded, which may underestimate their incidence. These factors should be considered when interpreting our results and warrant further prospective investigation.
In conclusion, ethanol with microcoil embolization is an effective and durable treatment for renal AML, achieving superior tumor volume reduction and treatment response compared to single-agent embolization. These findings highlight the effectiveness of this combination of embolic materials, supporting its use as the preferred approach for treating large or symptomatic AML.
Conflict of interest
Kun Yung Kim and Jae Hwan Lee, contributing editors of the Korean Journal of Interventional Radiology, were not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding
None.
Acknowledgments
None.
Author contributions
Conceptualization: KYK, YMH, JHL. Methodology: KYK, Minuk Kim, CJY, JHL. Investigation: KYK, MK, SHY, YMH, JHL. Data curation: KYK, MK. Formal analysis: KYK, MK. Supervision: CJY, CHL, YMH, JHL. Writing—original draft: KYK. Writing—review & editing: all authors. All authors read and approved the final manuscript.
Data availability statement
The datasets generated and/or analyzed during the current study are not publicly available due to institutional and privacy restrictions but are available from the corresponding authors upon reasonable request.
Fig. 1.Flow diagram of patient selection and inclusion in the study. AML, angiomyolipoma; TAE, transcatheter arterial embolization; eGFR, estimated glomerular filtration rate; PVA, polyvinyl alcohol.
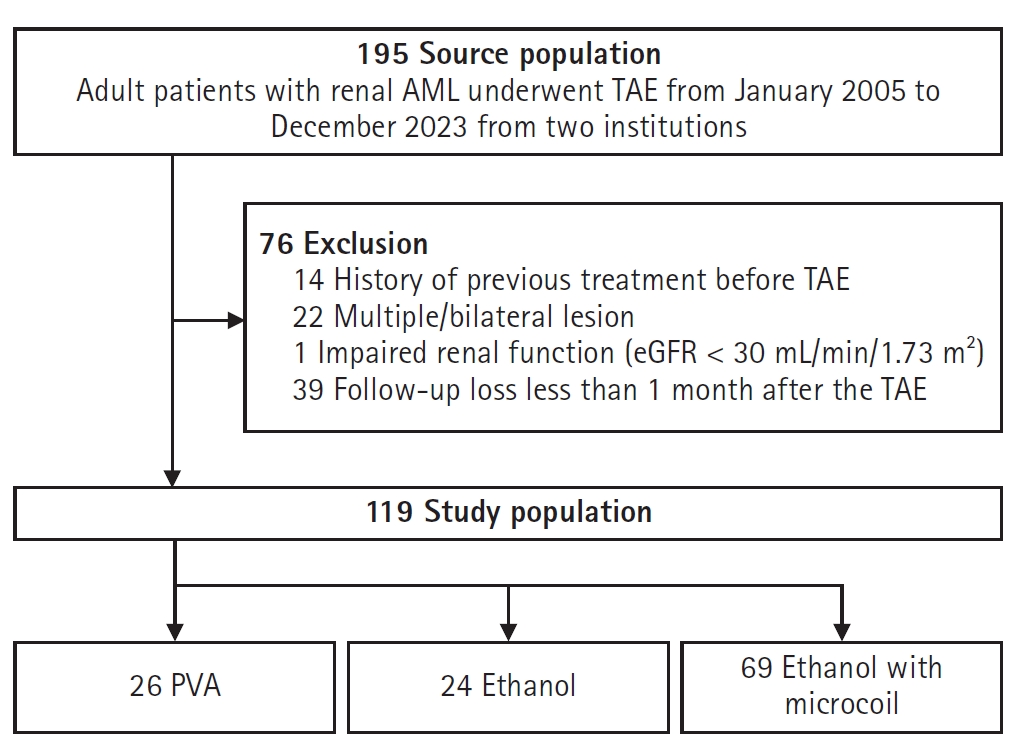
Table 1.Patient demographics before and after IPTW
Table 1.
|
Parameter |
Before IPTW
|
p-value |
SMD |
After IPTW
|
p-value |
SMD |
|
Ethanol |
Ethanol + microcoil |
PVA |
Ethanol |
Ethanol + microcoil |
PVA |
|
No. |
24 |
69 |
26 |
|
– |
47 |
42 |
30 |
– |
– |
|
Women (%) |
41.7 |
52.5 |
65.4 |
0.333 |
0.23 |
– |
– |
– |
– |
– |
|
Age (years) |
47.6 ± 12.8 |
50.9 ± 14.5 |
52.0 ± 16.5 |
0.334 |
0.18 |
– |
– |
– |
– |
– |
|
Baseline long diameter (mm) |
53.4 ± 19.6 |
61.9 ± 25.7 |
77.2 ± 27.2 |
0.001 |
1.02 |
63.5 ± 23.6 |
65.4 ± 28.6 |
66.0 ± 20.7 |
0.85 |
0.05 |
|
Baseline volume (cm3) |
93.5 ± 133.8 |
132.4 ± 172.6 |
281.1 ± 315.9 |
0.001 |
1.15 |
136.8 ± 184.9 |
159.5 ± 202.7 |
169.8 ± 217.1 |
0.72 |
0.04 |
Table 2.Tumor volume reduction between ethanol (ethanol and ethanol plus microcoil) and PVA before and after IPTW adjustment
Table 2.
|
Parameter |
Before IPTW
|
After IPTW
|
|
PVA |
Ethanol |
p-value |
PVA |
Ethanol |
Adjusted mean difference (ethanol – PVA) |
p-value |
|
Tumor volume (% of baseline) |
|
|
|
|
|
|
|
|
6-Month follow-up |
70.0 |
43.8 |
0.003 |
68.7 |
44.8 |
–23.9 |
0.002 |
|
12-Month follow-up |
49.2 |
25.1 |
0.018 |
48.9 |
25.8 |
–23.1 |
0.001 |
|
24-Month follow-up |
41.2 |
25.6 |
0.200 |
40.1 |
26.1 |
–14.0 |
0.266 |
|
≥50% Tumor reduction (response rate, %) |
69.2 |
87.1 |
0.031 |
78.4 |
92.1 |
OR, 2.86 |
0.043 |
|
Recurrence rate (%) |
15.4 |
12.9 |
0.743 |
14.8 |
13.3 |
OR, 0.88 |
0.844 |
Table 3.Tumor volume reduction between ethanol and ethanol plus microcoil before and after IPTW adjustment
Table 3.
|
Parameter |
Before IPTW
|
After IPTW
|
|
Ethanol only |
Ethanol + microcoil |
p-value |
Ethanol only |
Ethanol + microcoil |
Effect (microcoil vs. ethanol only) |
p-value |
|
Tumor volume (% of baseline) |
|
|
|
|
|
|
|
|
6-Month follow-up |
46.6 |
42.8 |
0.621 |
47.3 |
42.1 |
–5.2 |
0.510 |
|
12-Month follow-up |
25.4 |
25.0 |
0.936 |
27.2 |
24.7 |
–2.5 |
0.669 |
|
24-Month follow-up |
23.5 |
26.3 |
0.736 |
25.9 |
26.3 |
0.4 |
0.977 |
|
≥50% Tumor reduction (response rate, %) |
75.0 |
91.3 |
0.040 |
73.8 |
91.3 |
OR, 3.73 |
0.038 |
|
Recurrence rate (%) |
29.2 |
7.2 |
0.006 |
30.2 |
7.1 |
OR, 0.18 |
0.008 |
Table 4.Univariable and multivariable analyses of predictors for favorable treatment response
Table 4.
|
Parameter |
Univariable analysis |
Multivariable analysis |
|
OR (95% CI) |
p-value |
aOR (95% CI) |
p-value |
|
Embolic material type |
|
|
|
|
|
PVA |
|
0.026 |
|
0.026 |
|
Ethanol |
1.333 (0.384–4.625) |
0.650 |
1.333 (0.384–4.625) |
0.650 |
|
Ethanol with microcoil |
4.667 (1.433–15.203) |
0.011 |
4.667 (1.433–15.203) |
0.011 |
|
Age (years) |
0.974 (0.939–1.010) |
0.148 |
- |
- |
|
Female sex |
0.406 (0.111–1.489) |
0.174 |
- |
- |
|
Initial tumor size |
|
|
|
|
|
Long diameter (mm) |
0.990 (0.973–1.007) |
0.256 |
- |
- |
|
Short diameter (mm) |
0.984 (0.960–1.008) |
0.196 |
- |
- |
|
Z-axis diameter (mm) |
0.989 (0.975–1.004) |
0.146 |
- |
- |
|
Tumor volume (cm3) |
1.000 (1.000–1.000) |
0.474 |
- |
- |
References
- 1. Katabathina VS, Vikram R, Nagar AM, Tamboli P, Menias CO, Prasad SR. Mesenchymal neoplasms of the kidney in adults: imaging spectrum with radiologic-pathologic correlation. Radiographics. 2010;30:1525-1540. https://doi.org/10.1148/rg.306105517
- 2. Fernández-Pello S, Hora M, Kuusk T, Tahbaz R, Dabestani S, Abu-Ghanem Y, et al. Management of sporadic renal angiomyolipomas: a systematic review of available evidence to guide recommendations from the European Association of Urology Renal Cell Carcinoma Guidelines Panel. Eur Urol Oncol. 2020;3:57-72. https://doi.org/10.1016/j.euo.2019.04.005
- 3. Nason GJ, Morris J, Bhatt JR, Richard PO, Martin L, Ajib K, et al. Natural history of renal angiomyolipoma favors surveillance as an initial approach. Eur Urol Focus. 2021;7:582-588. https://doi.org/10.1016/j.euf.2020.06.004
- 4. Yamakado K, Tanaka N, Nakagawa T, Kobayashi S, Yanagawa M, Takeda K. Renal angiomyolipoma: relationships between tumor size, aneurysm formation, and rupture. Radiology. 2002;225:78-82. https://doi.org/10.1148/radiol.2251011477
- 5. Chatziioannou A, Gargas D, Malagari K, Kornezos I, Ioannidis I, Primetis E, et al. Transcatheter arterial embolization as therapy of renal angiomyolipomas: the evolution in 15 years of experience. Eur J Radiol. 2012;81:2308-2312. https://doi.org/10.1016/j.ejrad.2011.06.003
- 6. Vaggers S, Rice P, Somani BK, Veeratterapillay R, Rai BP. Evidence-based protocol-led management of renal angiomyolipoma: a review of literature. Turk J Urol. 2021;47(Suppl 1):S9-S18. https://doi.org/10.5152/tud.2020.20343
- 7. Flum AS, Hamoui N, Said MA, Yang XJ, Casalino DD, McGuire BB, et al. Update on the diagnosis and management of renal angiomyolipoma. J Urol. 2016;195:834-846. https://doi.org/10.1016/j.juro.2015.07.126
- 8. Kato H, Kuwatsuru R, Inoue T, Okada S, Aida M, Yamashiro Y. Superselective transcatheter arterial embolization for large unruptured renal angiomyolipoma in lymphangioleiomyomatosis. J Vasc Interv Radiol. 2018;29:958-965. https://doi.org/10.1016/j.jvir.2017.11.003
- 9. Hocquelet A, Cornelis F, Le Bras Y, Meyer M, Tricaud E, Lasserre AS, et al. Long-term results of preventive embolization of renal angiomyolipomas: evaluation of predictive factors of volume decrease. Eur Radiol. 2014;24:1785-1793. https://doi.org/10.1007/s00330-014-3244-4
- 10. Lee W, Kim TS, Chung JW, Han JK, Kim SH, Park JH. Renal angiomyolipoma: embolotherapy with a mixture of alcohol and iodized oil. J Vasc Interv Radiol. 1998;9:255-261. https://doi.org/10.1016/s1051-0443(98)70266-0
- 11. Jung Y, Choi MJ, Kim BM, Kim YM, Seo Y. Transarterial embolization for sporadic renal angiomyolipoma: patient selection and technical considerations for optimal therapeutic outcomes. Taehan Yongsang Uihakhoe Chi. 2022;83:559-581. https://doi.org/10.3348/jksr.2021.0120
- 12. Lee S, Park HS, Hyun D, Cho SK, Park KB, Shin SW, et al. Radiologic and clinical results of transarterial ethanol embolization for renal angiomyolipoma. Eur Radiol. 2021;31:6568-6577. https://doi.org/10.1007/s00330-021-07831-y
- 13. Urbano J, Paul L, Cabrera M, Alonso-Burgos A, Gomez D. Elective and emergency renal angiomyolipoma embolization with ethylene vinyl alcohol copolymer: feasibility and initial experience. J Vasc Interv Radiol. 2017;28:832-839. https://doi.org/10.1016/j.jvir.2017.01.017
- 14. Villalta JD, Sorensen MD, Durack JC, Kerlan RK, Stoller ML. Selective arterial embolization of angiomyolipomas: a comparison of smaller and larger embolic agents. J Urol. 2011;186:921-927. https://doi.org/10.1016/j.juro.2011.04.082
- 15. Wang MQ, Duan F, Zhang H, Zhang JL, Fu J, Ye HY, et al. Comparison of polyvinyl alcohol versus combination of lipiodol-bleomycin emulsion and NBCA-lipiodol emulsion for renal angiomyolipoma embolization: a prospective randomized study. AJR Am J Roentgenol. 2023;220:873-883. https://doi.org/10.2214/AJR.22.28587
- 16. Bissler JJ, Kingswood JC, Radzikowska E, Zonnenberg BA, Frost M, Belousova E, et al. Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet. 2013;381:817-824. https://doi.org/10.1016/S0140-6736(12)61767-X
- 17. Murray TE, Doyle F, Lee M. Transarterial embolization of angiomyolipoma: a systematic review. J Urol. 2015;194:635-639. https://doi.org/10.1016/j.juro.2015.04.081
- 18. Duffy M, Deshwal A, Donnelly R, Deshwal A, Mau A, Modi R, et al. Complication rates and effectiveness of renal angiomyolipoma embolisation: a systematic review and meta-analysis. Cardiovasc Intervent Radiol. 2026;49:195-209. https://doi.org/10.1007/s00270-025-04205-5
- 19. Shen Y, Han Q, Wang D, Su L, Wen M, Fan X, et al. Coil-assisted ethanol embolization of traumatic arteriovenous fistulas: a 10-year retrospective study. Front Cardiovasc Med. 2024;11:1449480. https://doi.org/10.3389/fcvm.2024.1449480
 , Minuk Kim2
, Minuk Kim2 , Chang Jin Yoon1,3,4
, Chang Jin Yoon1,3,4 , Chong-ho Lee1
, Chong-ho Lee1 , Sung-Hwan Yoon5
, Sung-Hwan Yoon5 , Young-Min Han6,7,*
, Young-Min Han6,7,* , Jae Hwan Lee1,3,4,*
, Jae Hwan Lee1,3,4,*