Abstract
-
Purpose
Tumor location influences the effectiveness and safety of RFA. This study evaluated RFA outcomes as first-line therapy for HCC <3 cm, focusing on tumor location impact.
-
Materials and Methods
In this retrospective cohort study, 281 patients with newly diagnosed HCC <3 cm in up to three lesions treated with RFA between 2003 and 2019 were analyzed. The tumor location was categorized as superficial (outer third), mid-portion (middle third), or deep (near vena cava), using an imaginary line from the liver surface to the vena cava. Perivascular tumors were defined as those abutting portal or hepatic veins. Recurrence-free survival (RFS) among location groups was compared with risk factors analyzed via Cox regression.
-
Results
Patients (mean age, 61.1 ± 11.1 years) were predominantly male (73.3%), hepatitis B virus surface antigen–positive (66.2%), and of Child-Pugh class A (97.5%). Deep tumors had shorter RFS than superficial/mid-portion tumors (HR, 1.87; 95% CI, 1.20 to 2.93; p = 0.005), as did perivascular versus non-perivascular tumors (HR, 1.87; 95% CI, 1.16 to 3.00; p = 0.008). Group C (deep + perivascular, n = 10) had shorter RFS than group A (no risk factors: HR, 3.12; 95% CI, 1.50 to 6.45; p = 0.002) and group B (one risk factor: HR, 1.59; 95% CI, 1.05 to 2.40; p = 0.028). Multivariable analysis identified tumor depth, perivascular location, size >2 cm, creatinine, and prothrombin time as independent predictors of shorter RFS.
-
Conclusion
Tumor depth and proximity to vasculature independently predict RFS in small HCCs treated with RFA, highlighting the role of tumor location in determining patient prognosis.
-
Keywords: Hepatocellular carcinoma; Radiofrequency ablation; Prognosis; Local neoplasm recurrence; Neoplasms by site
Introduction
RFA is an established first-line treatment for very early (stage 0) and early (stage A) HCC [
1,
2]. The anatomical tumor location is pivotal in determining not only the RFA feasibility but also its clinical outcomes [
3-
6]. Tumors located adjacent to extrahepatic organs or major intrahepatic vessels present unique challenges and are often considered relative contraindications owing to the elevated risk of damaging surrounding tissues [
7,
8]. RFA efficacy is significantly reduced in perivascular HCCs due to the “heat sink” effect, where blood flow in nearby vessels dissipates heat from the ablation zone, limiting the effectiveness of the procedure and leading to lower overall survival (OS) rates than surgical options [
3,
4]. Despite these limitations, current international guidelines do not yet integrate anatomical considerations in RFA treatment recommendations, leaving a gap in optimizing treatment for these cases [
2,
9].
Challenges exist for RFA required at specific tumor locations. RFA is typically performed percutaneously for minimal invasiveness; however, certain tumor locations complicate the procedure [
4,
8,
10]. For superficial or subcapsular tumors, concerns arise regarding the potential for inadequate tumor margin and the risk of tumor seeding and recurrence [
11-
13]. Deep-seated HCCs, including caudate lobe lesions, further complicate detection and access, making precise targeting during surgery or locoregional treatments challenging [
14-
16]. These cases are often associated with higher rates of surgical complications and poorer outcomes [
14-
18].
Previous studies on RFA for challenging tumor locations have focused mainly on technical feasibility in small patient cohorts [
19-
21] lacking detailed outcome analyses stratified by specific anatomical tumor locations in larger patient samples. Therefore, this study aimed to evaluate RFA outcomes, accounting for anatomical tumor location, which influences treatment efficacy and patient prognosis. By quantifying these effects, this analysis seeks to inform guidelines and aid in developing targeted treatment protocols for HCC based on anatomical considerations.
Materials and Methods
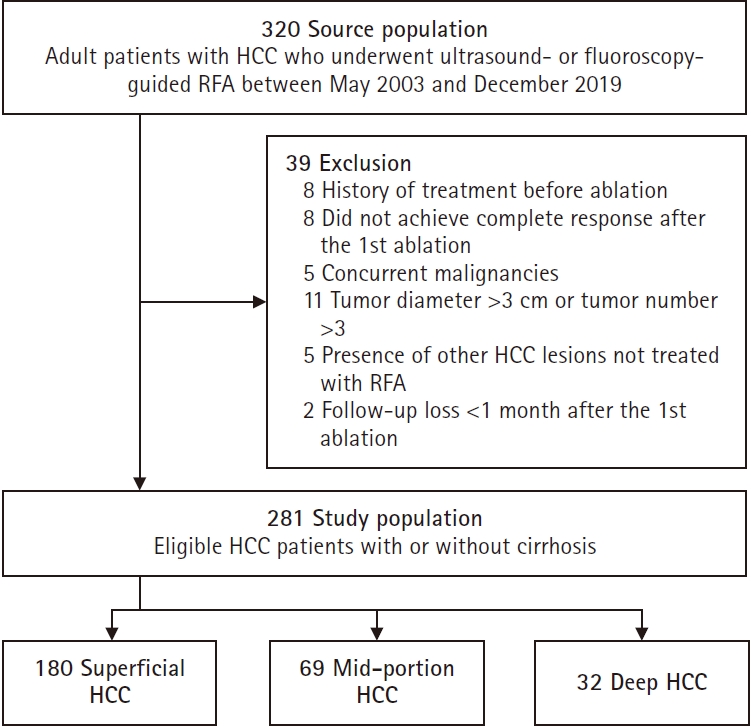
This retrospective cohort study was conducted at a single tertiary academic center, with approval from the Institutional Review Board (14/10/2020) and a waiver of informed consent. A total of 320 treatment-naïve patients with HCC at our institution between May 2003 and December 2019 were initially screened. After applying the eligibility criteria, including exclusion of patients with prior HCC treatment (n = 8), 281 treatment-naïve patients were included in the final analysis. The diagnostic criteria for HCC were based on the international guidelines at the time of diagnosis [
22,
23]. The inclusion criteria were: [
1] newly diagnosed HCC confirmed through international guidelines or biopsy in cases of indeterminate lesions; [
2] a maximum of three tumors, each <3 cm in diameter; [
3] non-suitability for surgical resection; [
4] RFA as the initial therapy; and [
5] complete response achieved after a single RFA session, defined as the absence of contrast enhancement in the ablated area during the arterial phase, uniform low attenuation within the ablation zone, and a minimum 0.5–1.0 cm ablative margin of non-enhancing liver tissue surrounding the tumor during the portal venous phase [
24]. Exclusion criteria included prior HCC treatment (n = 8), incomplete response post-first ablation (n = 8), presence of extrahepatic malignancies (n = 5), tumors of ≥3 cm (n = 11), concurrent HCC lesions untreated by RFA (n = 5), and lack of follow-up after RFA (n = 2). Following these criteria, 281 patients were included in the final analysis (
Fig. 1).
Based on previous studies [
3,
25] tumor depth was categorized as follows (
Supplemental Fig. 1): an imaginary line was drawn from the IVC through the tumor center to the nearest liver surface. The tumor depth was classified as superficial, middle, or deep according to the position of the tumor center along this line. Tumors with their center in the distal third, closer to the IVC, were classified as deep. A perivascular location was defined as tumors abutting the 1st or 2nd-degree branches of the portal or hepatic vein [
4]. Two radiologists, blinded to treatment outcomes, independently reviewed and classified all tumors by consensus based on pre-treatment CT or MRI scans.
All RFA procedures were performed on an inpatient basis by one of two radiologists using local anesthesia with conscious sedation. A 17-gauge internally cooled electrode with a manually adjustable active tip (0.5‒3 cm; Viva, Starmed, Goyang, Korea) was used for the procedure. Ultrasound guidance facilitated accurate targeting, with intraprocedural monitoring to confirm an adequate peritumoral ablation margin.
For tumors not clearly visible on ultrasound, contrast-enhanced ultrasound, and intraprocedural cone-beam CT scanning were performed in conjunction with RFA under ultrasonography and fluoroscopy guidance, following previously established protocols [
26]. In brief, ultrasound contrast agent SonoVue (sulfur hexafluoride microbubbles; Bracco Imaging, Milan, Italy) was initially administered to enhance visualization of the sonographically invisible lesion. Once the lesion was identified, RFA needle placement was performed under ultrasound guidance. Following this, intra-procedural cone-beam CT scanning was conducted to verify the position of the needle, allowing comparison with pre-procedural CT or MRI images to ensure precise placement within the tumor [
26,
27].
Clinical data for the study group were collected from electronic medical records and included age, sex, HCC etiology, complete blood count, alpha-fetoprotein blood levels, serum aspartate aminotransferase, alanine aminotransferase, albumin, total bilirubin, prothrombin time (PT) as international normalized ratio, creatinine, Child-Pugh score and classification, tumor size, number, location, distribution, and the presence of macrovascular invasion or distant metastasis.
Recurrence was defined as any radiologically confirmed reappearance of HCC after complete response to the index RFA, including local tumor progression, intrahepatic distant recurrence, and extrahepatic recurrence. The primary outcome was recurrence-free survival (RFS), and the secondary outcome was time to progression (TTP) and OS. RFS was defined as the time from the date of RFA procedure to the first recurrence observed on cross-sectional imaging (CT or MRI), death, or the end of follow-up. TTP was defined as the interval from the date of RFA procedure to the date of recurrence confirmed on imaging. OS was defined as the interval from the date of RFA procedure to the date of death. Patients were followed up until December 2022 or for up to 5 years. Mortality data were confirmed through Statistics Korea (governmental census bureau), and the cause of death was verified using medical records.
Statistical Analysis
Baseline patient characteristics were compared using the chi-square test for categorical variables and the t-test for continuous variables. RFS and TTP were estimated using the Kaplan-Meier method and compared with the log-rank test. A Cox proportional hazards model was used in both univariable and multivariable analyses to assess HR and 95% CI for RFS.
To address significant baseline differences, propensity score (PS) matching was applied, reducing the influence of potential confounders and improving the predictive value of tumor location estimates. Nearest-neighbor matching with a caliper of 0.1 was used to match patients in a 1:2 ratio. All p-values were two-sided, with values <0.05 considered statistically significant. Statistical analyses were conducted using SPSS version 21 (IBM Corp., Armonk, NY, USA) and R version 4.0.4 (
http://cran.r-project.org/).
This study was approved by the Institutional Review Board of Seoul National University Bundang Hospital (approval No. B-1912/580-104). The institutional review board waived the need for informed consent due to the retrospective nature of the study. Consent for publication was obtained for every person’s data included in the study.
Results
The baseline characteristics of patients undergoing RFA by tumor depth are shown in
Table 1. Among the 281 patients, the mean age was 61.1 ± 11.1 years, with 73.3% being male, 66.2% hepatitis B virus surface antigen–positive, and 97.5% in Child-Pugh class A. Tumor characteristics, including maximal tumor diameter (17.2 mm, 18.1 mm, and 17.4 mm for superficial, middle, and deep HCC, respectively) and the proportion of patients with a single tumor (93.9%, 91.3%, and 96.6%, respectively), did not differ significantly among groups.
Anatomical distribution showed that superficial and middle HCCs were absent in segment 1, while deep HCCs were not located in segments 3 or 5. Perivascular location was progressively more frequent from superficial to deep HCCs (10.5% for superficial, 18.8% for middle, and 31.3% for deep; p = 0.007). Similarly, Child-Pugh class B status increased with tumor depth (0% for superficial, 2.9% for middle, and 15.6% for deep HCC; p < 0.001).
TTP, RFS, and OS by Tumor Depth before PS Matching
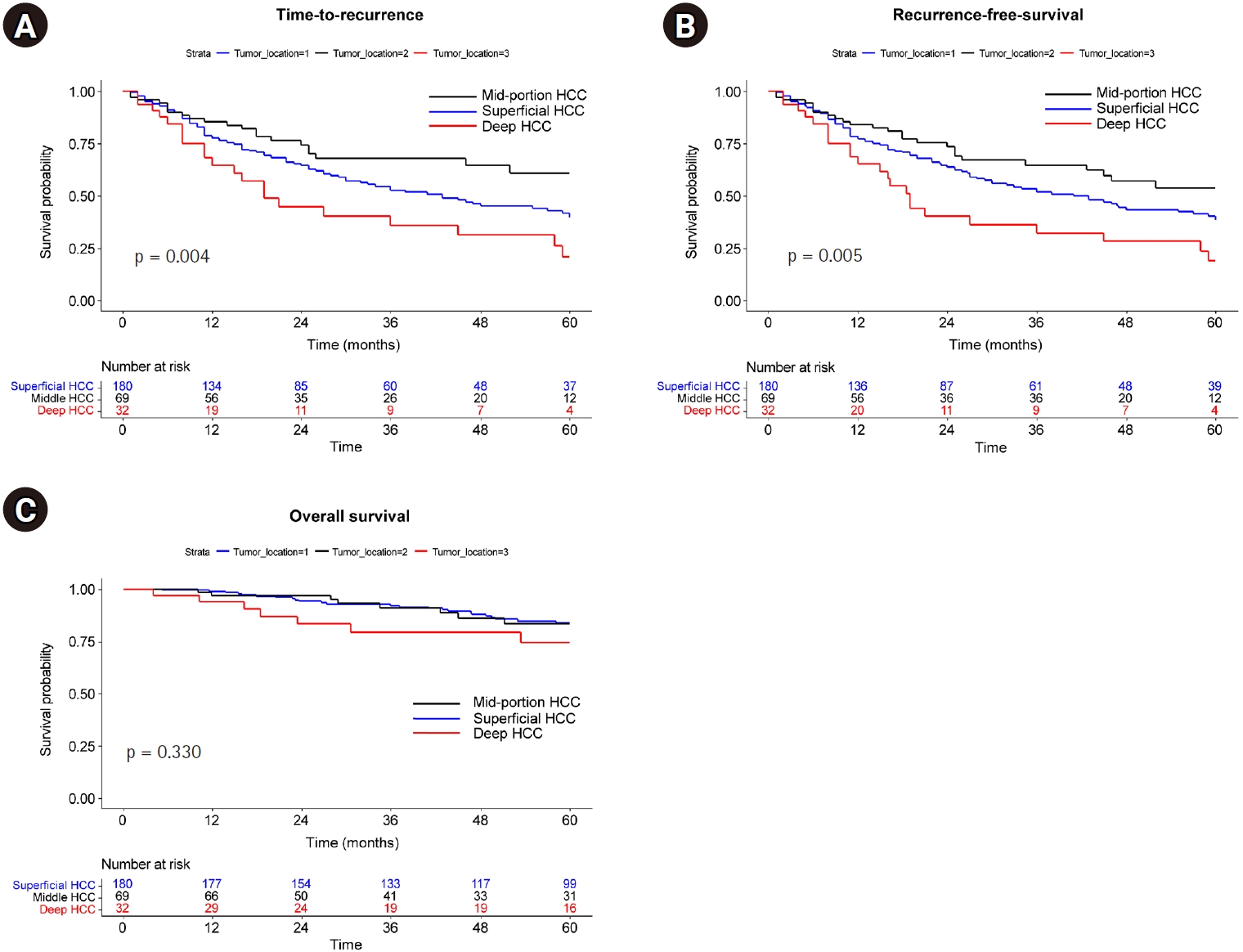
The median follow-up for study patients was 60 months, during which 128 recurrences and 39 deaths were observed. TTP was shortest in deep HCCs, followed by superficial and middle HCCs (median, not reached [NR] vs. 43 months vs. 19 months; p = 0.004) (
Fig. 2A). RFS similarly decreased in the order of middle, superficial, and deep HCCs (median, NR vs. 41 months vs. 19 months; p = 0.004 for trend) (
Fig. 2B). However, OS was not significantly different among the three groups (
Fig. 2C).
Multivariable analysis identified deep HCC as an independent risk factor for shorter RFS (
Table 2), with an HR of 2.16 (95% CI, 1.20 to 3.89; p = 0.010) compared to middle HCCs, while superficial HCCs showed no significant association (HR, 1.44; 95% CI, 0.92 to 2.25; p = 0.112). Additional covariates independently linked to shorter RFS included elevated creatinine (HR, 1.14; 95% CI, 1.00 to 1.29; p = 0.042), perivascular location (HR, 1.65; 95% CI, 1.01 to 2.72; p = 0.048), and prolonged PT (HR, 6.84; 95% CI, 1.92 to 24.35; p = 0.003).
Patients with perivascular tumors had shorter TTP (median, 22 months vs. 52 months; log-rank p = 0.008), RFS (median, 22 months vs. 46 months; p = 0.006), and OS (p = 0.026) compared to those with non-perivascular tumors (
Supplemental Fig. 2).
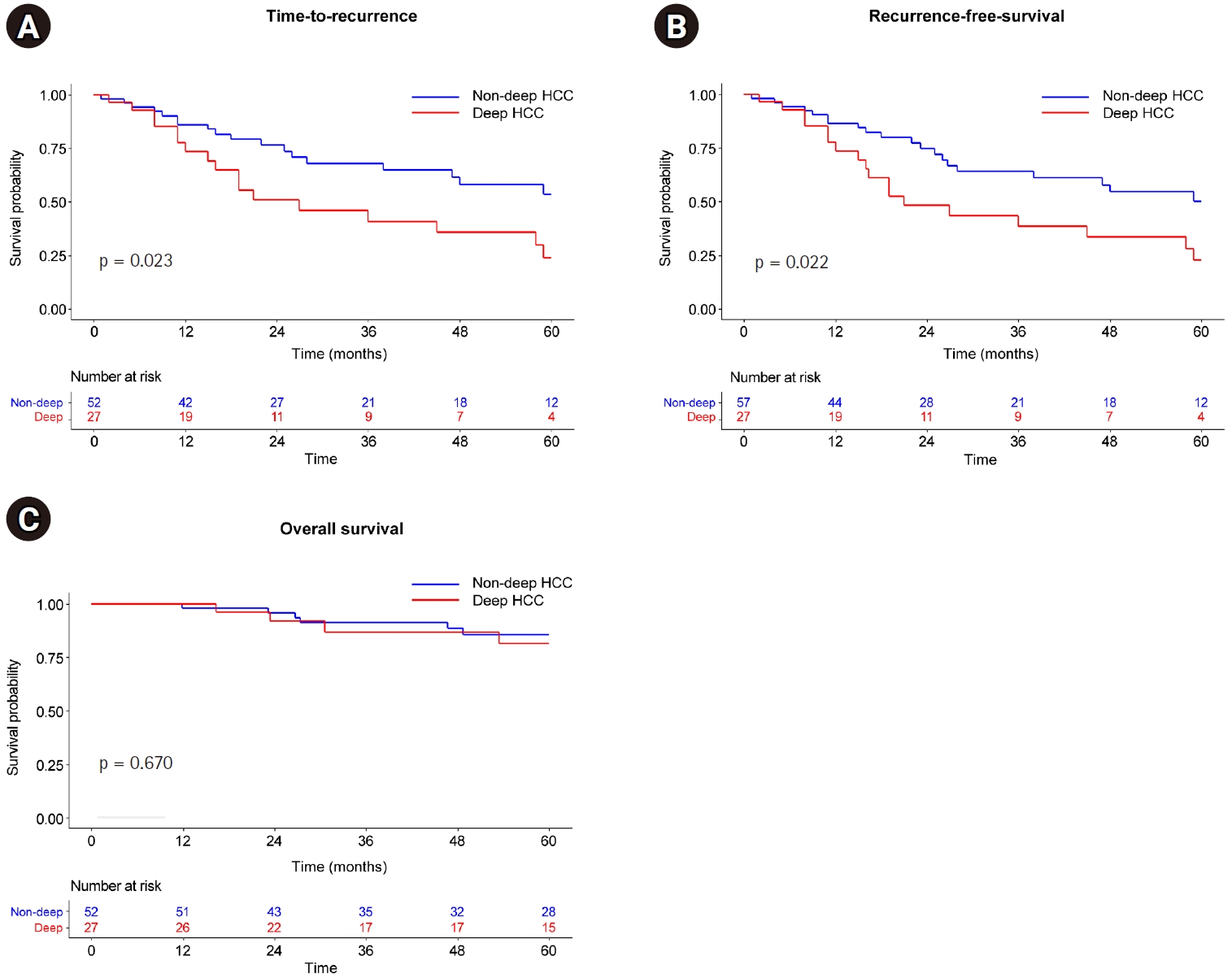
Because superficial and middle HCC did not show statistically significant differences in multivariable analysis, they were divided into deep HCC and non-deep HCC groups and PS matching was performed. PS matching yielded 27 pairs (ratio 1:2) of patients from the deep and non-deep HCC groups (
Supplemental Table 1), with no significant differences between the matched cohorts. In this matched analysis, the deep HCC group demonstrated a higher risk of shorter TTP (HR, 2.11; 95% CI, 1.08 to 4.09; p = 0.023) and RFS (HR, 2.07; 95% CI, 1.09 to 3.91; p = 0.022) than those in the non-deep HCC group (
Fig. 3A,
B); however, the OS of patients with deep HCC did not differ significantly from those with non-deep HCC (p = 0.670) (
Fig. 3C).
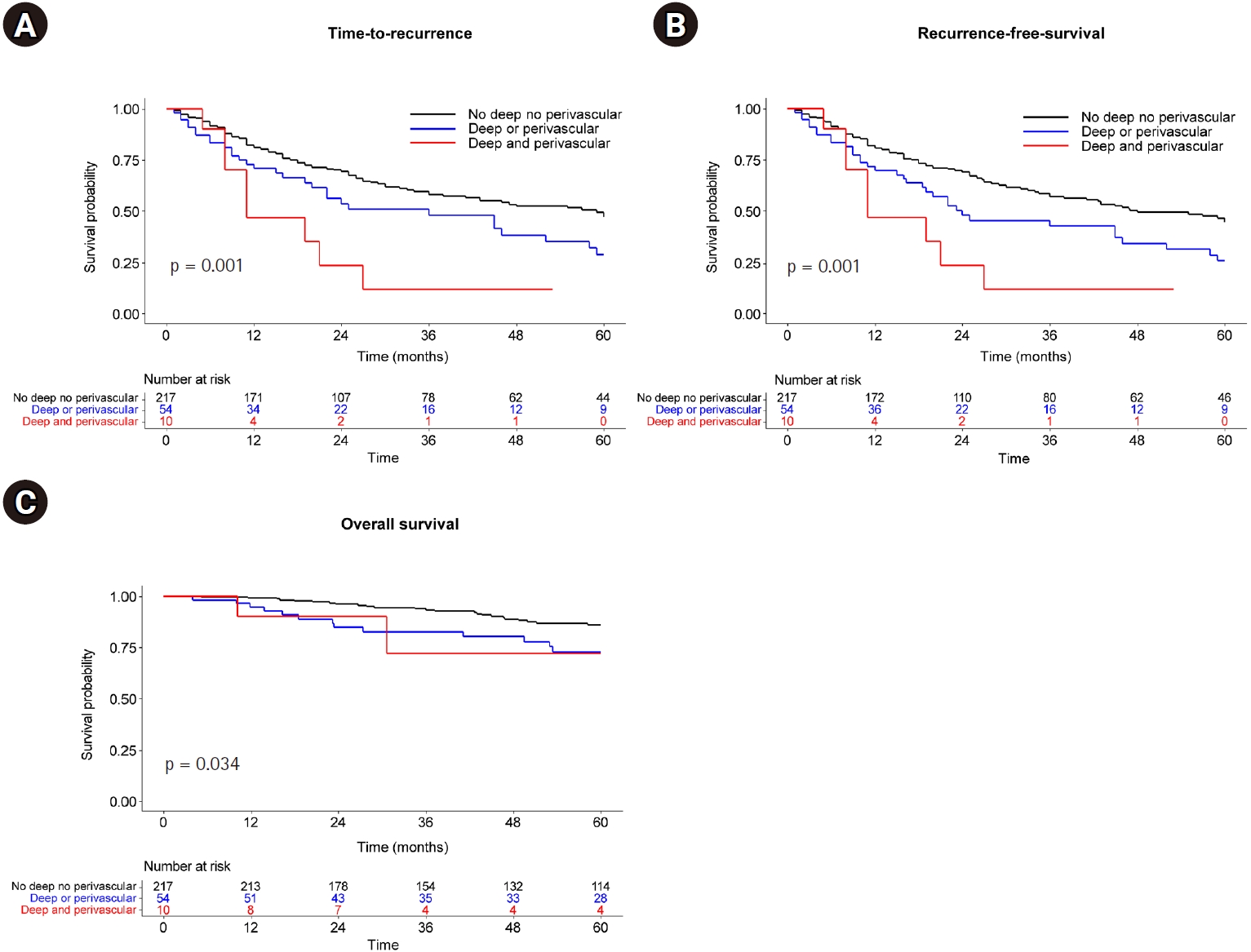
Patients were stratified into three groups based on the presence of two anatomical risk factors (deep HCCs and perivascular location): non-deep, non-perivascular group (n = 217), deep or perivascular group (n = 54), and deep and perivascular group (n = 10).
TTP, RFS, and OS showed a significantly favorable prognosis in the order of the non-deep, non-perivascular; deep or perivascular; and deep and perivascular groups (
Fig. 4). Moreover, multivariable Cox analysis (
Table 2) demonstrated that the number of anatomical risk factors independently predicted shorter RFS (HR, 1.60; 95% CI, 1.07 to 2.40; p = 0.024 for deep or perivascular HCC; HR, 2.41; 95% CI, 1.08 to 5.38, p = 0.031 for deep and perivascular HCC with reference to non-deep, non-perivascular HCC). Additional factors associated with shorter RFS included maximal tumor size >2 cm (HR, 1.62; 95% CI, 1.12 to 2.34; p = 0.011), elevated creatinine (HR, 1.14; 95% CI, 1.01 to 1.29; p = 0.042), and prolonged PT (HR, 20.51; 95% CI, 4.20 to 100.26; p < 0.001).
Discussion
In this study, patients with deep-seated HCCs and perivascular tumor locations demonstrated significantly shorter RFS and TTP than patients with tumors in less challenging locations. Deep HCCs, specifically, were identified as independent predictors of shorter RFS and TTP. Stratification based on anatomical risk factors showed that patients with both deep and perivascular tumors had the poorest outcomes, underscoring the influence of tumor location on therapeutic efficacy.
Tumor location has a profound impact on the effectiveness of curative therapies for HCC, as it directly affects procedural feasibility and oncologic outcomes [
3,
4,
6-
8,
10,
28]. Anatomically challenging locations, such as perivascular or subcapsular areas, complicate RFA due to technical difficulties in achieving adequate margins and risks of adjacent tissue damage [
4,
28]. For instance, tumors adjacent to large blood vessels experience the “heat sink” effect, leading to incomplete ablation and higher recurrence rates [
3,
4]. International guidelines do not yet account for tumor location in treatment planning for HCC [
9,
29]. However, emerging evidence, including this study, suggests that incorporating location-based considerations could enhance treatment personalization and improve outcomes.
Our findings indicate that deep-seated HCCs were found to be independent predictors of shorter RFS, which underscores the challenges of treating tumors located in these anatomically complex regions. While technical factors, such as achieving adequate ablation margins due to proximity to major blood vessels, contribute to these outcomes, biological and microenvironmental factors likely play a substantial role. Deep-seated tumors may have reduced immunologic surveillance, potentially enabling tumor cells to evade immune detection and increasing the risk of recurrence [
30]. In addition, the unique vascular microenvironment around deep HCCs might support faster tumor regrowth and micrometastasis formation, which standard imaging and local treatment modalities could fail to detect [
3,
4,
31].
These findings highlight the importance of pre-treatment assessment of tumor depth, as deeper tumors are likely to require modified or adjunctive therapeutic approaches, such as the integration of transarterial chemoembolization to complement RFA or microwave ablation [
5,
31,
32]. Clinically, the poorer prognosis for deep-seated HCCs suggests a need for heightened surveillance and consideration of alternative therapies for these patients [
21].
Stratifying patients based on anatomical risk factors, such as tumor depth and perivascular proximity, allows for a more nuanced understanding of prognosis post-RFA. In this study, patients grouped by these risk factors showed progressively worse RFS outcomes with increased anatomical complexity. This stratification system could aid clinicians in identifying high-risk patients, guiding treatment selection, and improving patient counseling.
This study has several limitations. As a retrospective, single-center study, it may be subject to inherent biases in patient selection and data collection. Second, the enrollment period spanned more than 15 years, during which RFA devices and image-guidance techniques evolved. Such temporal changes may have influenced treatment outcomes, particularly for deep or perivascular tumors that are more dependent on advanced guidance techniques. Therefore, the prognostic effect of anatomical location may not have been constant throughout the study period. Additionally, the modest sample size limits the generalizability of findings to broader populations. Future multi-center, prospective studies are needed to validate these results and further refine anatomical risk stratification in HCC management.
In conclusion, this study emphasizes that tumor location significantly impacts outcomes in HCC patients treated with RFA, particularly in deep and perivascular tumors linked to poorer prognosis. These findings support anatomical risk stratification to guide treatment decisions and suggest updates to clinical guidelines to account for tumor location in HCC management.
Conflict of interest
Kun Yung Kim and Jae Hwan Lee, editors of the Korean Journal of Interventional Radiology, were not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding
This work was supported by the Central Medical Service (CMS) Co., Ltd. Research Fund (Grant number B-2306-835-103).
Acknowledgments
We would like to thank Editage (http://www.editage.com) for editing and reviewing this manuscript for the English language.
Author contributions
Conceptualization: GHC, JHL. Data curation: GHC, KYK, MK. Formal analysis: GHC, SJL, JHL. Investigation: GHC, CHL, CJY. Funding acquisition: JHL. Methodology: GHC, CJY, JHL. Project administration: JHL. Resources: CJY, ESJ, JWK, SHJ. Software: SJL. Supervision: JHL. Validation: GHC, JHL. Visualization: GHC, SJL. Writing—original draft: GHC. Writing—review & editing: all authors.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplementary material
Fig. 1.
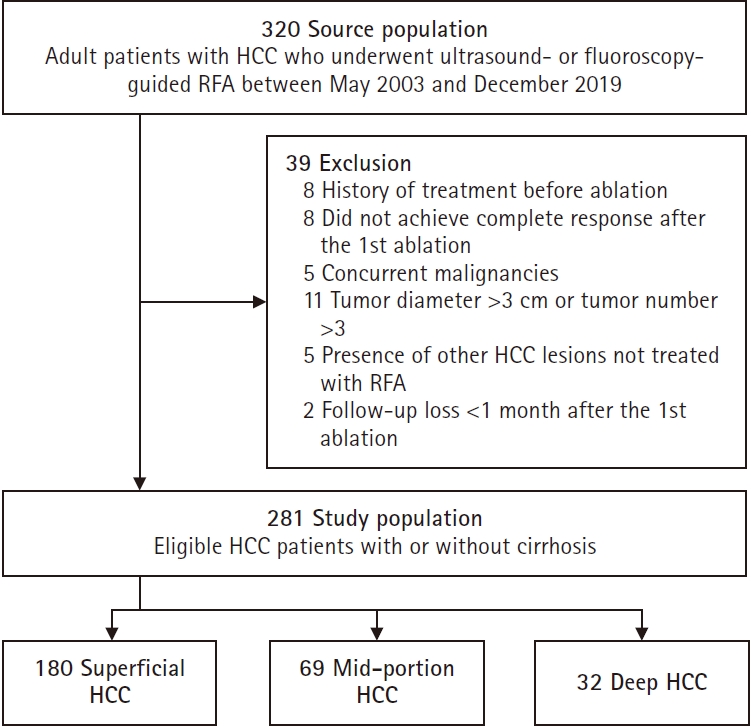
Fig. 2.Kaplan-Meier curve according to tumor depth. (A) Time to recurrence. (B) Recurrence-free survival. (C) Overall survival.
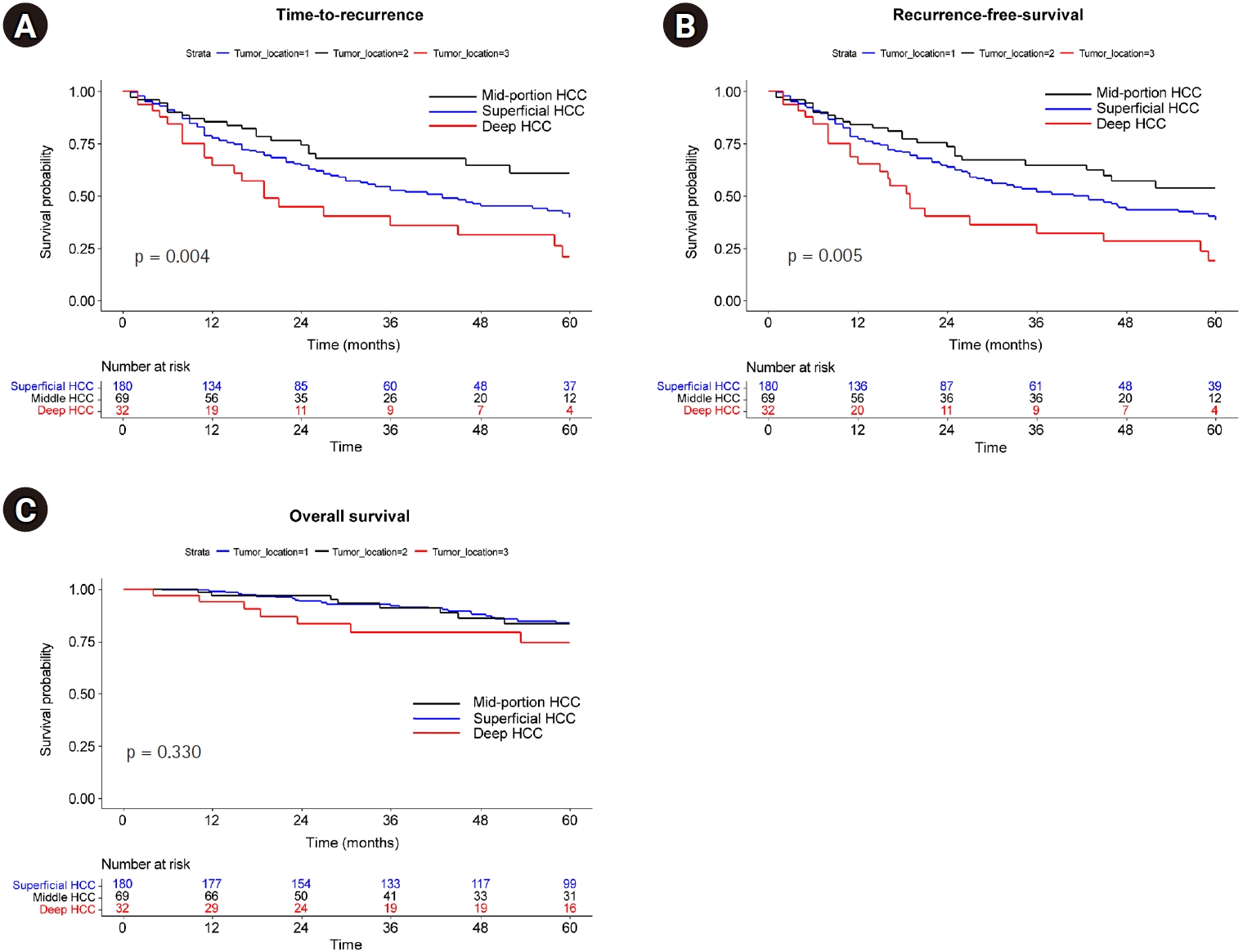
Fig. 3.Kaplan-Meier curve after propensity score matching according to depth. (A) Time to recurrence. (B) Recurrence-free survival. (C) Overall survival.
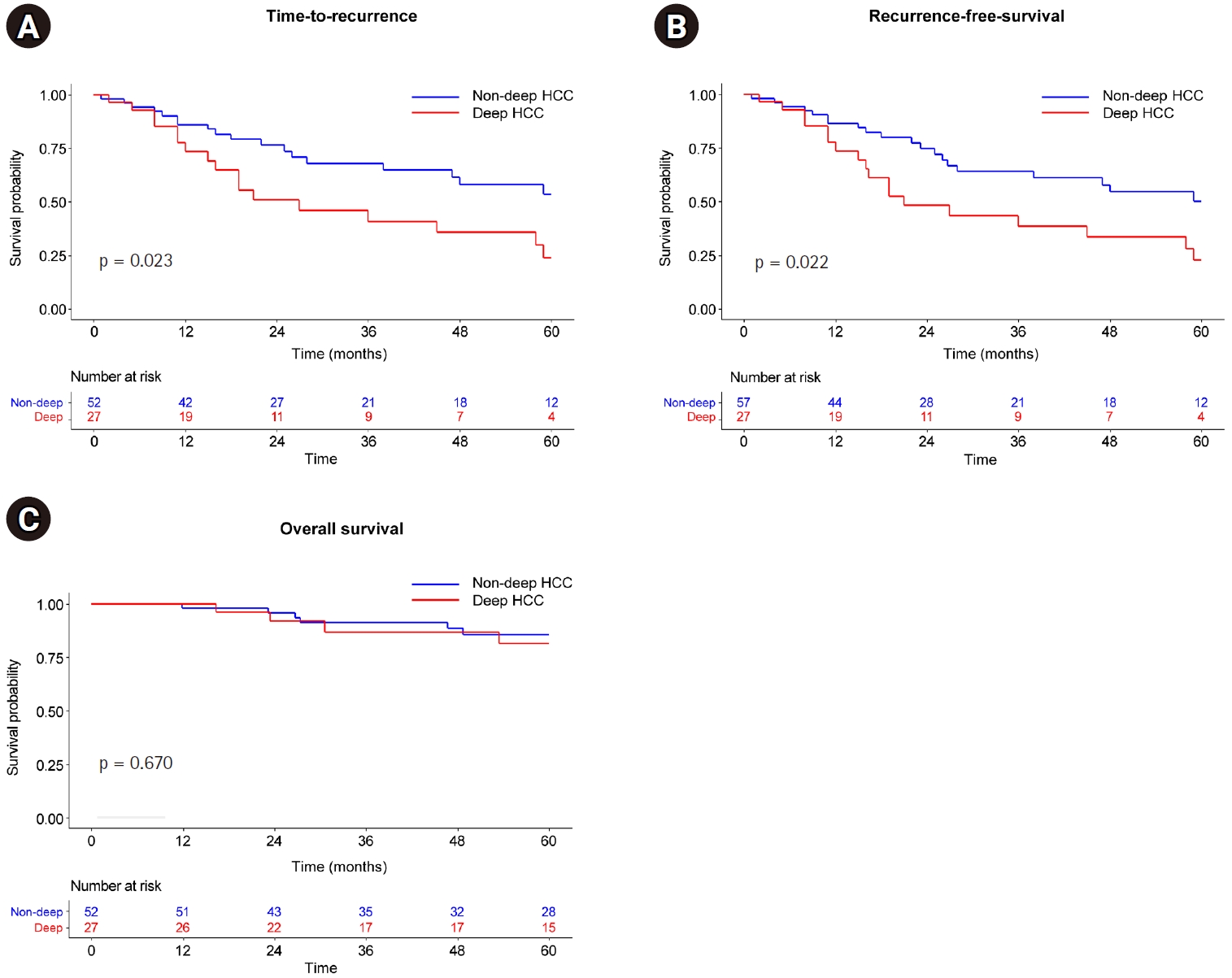
Fig. 4.Kaplan-Meier curve according to deep and/or perivascular location. (A) Time to recurrence. (B) Recurrence-free survival. (C) Overall survival.
Table 1.Baseline characteristics of HCC patients who received radiofrequency ablation
Table 1.
|
Superficial (n = 180) |
Mid (n = 69) |
Deep (n = 32) |
p-value |
|
Age (years) |
61.3 ± 11.2 |
61.6 ± 10.6 |
58.6 ± 11.2 |
0.405 |
|
Male sex |
131 (72.8) |
51 (73.9) |
24 (75.0) |
0.958 |
|
Etiology |
|
|
|
0.986 |
|
HBV/HCV |
120 (66.7)/21 (11.7) |
45 (65.2)/9 (13.0) |
21 (65.6)/3 (9.4) |
|
|
Alcohol/others |
36 (20.0)/4 (1.7) |
12 (17.4)/3 (4.3) |
8 (25.0)/0 (0) |
|
|
Maximal tumor diameter (D) (mm) |
17.2 ± 5.0 |
18.1 ± 5.4 |
17.4 ± 5.4 |
0.422 |
|
D < 20 |
137 (76.1) |
51 (73.9) |
23 (71.9) |
0.849 |
|
20 ≤ D < 30 |
43 (23.9) |
18 (26.1) |
9 (28.1) |
|
|
Tumor number |
|
|
|
|
|
1/2 |
169 (93.9)/11 (6.1) |
63 (91.3)/6 (8.7) |
31 (96.9)/1 (3.1) |
0.548 |
|
Tumor segment |
|
|
|
<0.001 |
|
1/2 |
0 (0)/5 (2.8) |
0 (0)/1 (1.4) |
5 (15.6)/1 (3.1) |
|
|
3/4 |
19 (10.6)/16 (8.9) |
4 (5.8)/5 (7.2) |
0 (0)/2 (6.2) |
|
|
5/6 |
23 (12.8)/56 (31.1) |
7 (10.1)/19 (27.5) |
0 (0)/7 (21.9) |
|
|
7/8 |
7 (3.9)/54 (30.0) |
7 (10.1)/26 (37.7) |
7 (21.9)/10(31.2) |
|
|
Perivascular location |
|
|
|
0.007 |
|
Portal vein/hepatic vein |
15 (8.3)/4 (2.2) |
11 (15.9)/2 (2.9) |
6 (18.8)/4 (12.5) |
|
|
Child-Pugh class B |
0 (0) |
2 (2.9) |
5 (15.6) |
<0.001 |
|
Laboratory findings |
|
|
|
|
|
WBC (/mm3) |
4.0 (3.9–6.0) |
5.2 (4.2–6.4) |
4.8 (3.8–6.0) |
0.104 |
|
PLT (×103/mm3) |
119 (83–164) |
124 (91–180) |
124 (90–159) |
0.351 |
|
ALT (IU/L) |
35 (23–53) |
32 (23–44) |
37 (28–50) |
0.147 |
|
ALP (IU/L) |
85 (68–105) |
88 (69–114) |
91 (75–127) |
0.215 |
|
Albumin (g/dL) |
4.0 (3.7–4.3) |
4.1 (3.7–4.4) |
4.0 (3.4–4.2) |
0.159 |
|
Total bilirubin (mg/dL) |
0.7 (0.5–1.1) |
0.9 (0.6–1.1) |
1.0 (0.6–1.4) |
0.001 |
|
Creatinine (mg/dL) |
0.8 (0.7–1.0) |
0.9 (0.7–1.0) |
0.9 (0.7–1.0) |
0.030 |
|
Prothrombin time (INR) |
1.09 (1.04–1.17) |
1.09 (1.02–1.15) |
1.11 (1.03–1.27) |
0.043 |
|
AFP (ng/dL) |
6.8 (3.4–38.2) |
6.2 (3.2–21.6) |
7.9 (3.3–34.0) |
0.644 |
Table 2.Multivariable Cox analysis for recurrence-free survival
Table 2.
|
Univariable analysis |
Multivariable analysis model 1 |
Multivariable analysis model 2 |
|
Hazard ratio |
p-value |
Hazard ratio |
p-value |
Hazard ratio |
p-value |
|
Tumor depth |
|
- |
|
- |
- |
- |
|
Mid HCC |
Reference |
|
Reference |
|
|
|
|
Superficial 1/3 HCC |
1.45 (0.93–2.26) |
0.099 |
1.44 (0.92–2.25) |
0.112 |
|
|
|
Deep 1/3 HCC |
2.48 (1.41–4.37) |
0.002 |
2.16 (1.20–3.89) |
0.010 |
|
|
|
Perivascular HCC |
1.87 (1.16–3.00) |
0.010 |
1.65 (1.01–2.72) |
0.048 |
|
|
|
Tumor location |
|
|
|
|
|
|
|
Non-deep, non-perivascular |
Reference |
- |
- |
- |
Reference |
- |
|
Deep or perivascular |
1.66 (1.12–2.46) |
0.011 |
- |
- |
1.60 (1.07–2.40) |
0.024 |
|
Deep and perivascular |
2.99 (1.45–6.18) |
0.003 |
- |
- |
2.41 (1.08–5.38) |
0.031 |
|
Maximal tumor diameter ≥ 20 mm |
1.51 (1.05–2.18) |
0.025 |
1.56 (1.07–2.26) |
0.020 |
1.62 (1.12–2.34) |
0.011 |
|
Two HCC |
1.28 (0.67–2.43) |
0.457 |
- |
- |
- |
- |
|
Age (years) |
1.00 (0.99–1.02) |
0.669 |
- |
- |
- |
- |
|
Male sex |
1.23 (0.83–1.81) |
0.308 |
- |
- |
- |
- |
|
PLT (×1,000/mm3) |
0.997 (0.994–1.000) |
0.055 |
0.999 (0.996–1.002) |
0.657 |
0.999 (0.996–1.002) |
0.608 |
|
ALT (IU/L) |
1.000 (0.996–1.003) |
0.884 |
- |
- |
- |
- |
|
Albumin (g/dL) |
0.60 (0.42–0.84) |
0.003 |
0.86 (0.57–1.31) |
0.483 |
0.88 (0.58–1.33) |
0.536 |
|
Total bilirubin (mg/dL) |
1.24 (0.95–1.61) |
0.113 |
- |
- |
- |
- |
|
Prothrombin time (INR) |
11.05 (3.90–31.28) |
<0.001 |
6.84 (1.92–24.35) |
0.003 |
20.51 (4.20–100.26) |
<0.001 |
|
Creatinine (mg/dL) |
1.13 (1.01–1.28) |
0.038 |
1.14 (1.00–1.29) |
0.042 |
1.14 (1.00–1.29) |
0.042 |
|
AFP > 20 ng/mL |
0.94 (0.64–1.36) |
0.725 |
- |
- |
- |
- |
References
- 1. Korean Liver Cancer Association (KLCA); National Cancer Center (NCC) Korea. 2022 KLCA-NCC Korea practice guidelines for the management of hepatocellular carcinoma. J Liver Cancer. 2023;23:1-120. https://doi.org/10.17998/jlc.2022.11.07
- 2. Reig M, Forner A, Rimola J, Ferrer-Fabrega J, Burrel M, Garcia-Criado A, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76:681-693. https://doi.org/10.1016/j.jhep.2021.11.018
- 3. Chen J, Peng K, Hu D, Shen J, Zhou Z, Xu L, et al. Tumor location influences oncologic outcomes of hepatocellular carcinoma patients undergoing radiofrequency ablation. Cancers (Basel). 2018;10:378. https://doi.org/10.3390/cancers10100378
- 4. Lee S, Kang TW, Cha DI, Song KD, Lee MW, Rhim H, et al. Radiofrequency ablation vs. surgery for perivascular hepatocellular carcinoma: propensity score analyses of long-term outcomes. J Hepatol. 2018;69:70-78. https://doi.org/10.1016/j.jhep.2018.02.026
- 5. Doyle A, Gorgen A, Muaddi H, Aravinthan AD, Issachar A, Mironov O, et al. Outcomes of radiofrequency ablation as first-line therapy for hepatocellular carcinoma less than 3 cm in potentially transplantable patients. J Hepatol. 2019;70:866-873. https://doi.org/10.1016/j.jhep.2018.12.027
- 6. Dou JP, Han ZY, Cheng ZG, Liu FY, Yu XL, Yu J, et al. The effect of tumor location on long-term results of microwave ablation for early-stage hepatocellular carcinoma. Abdom Radiol (NY). 2020;45:3923-3933. https://doi.org/10.1007/s00261-020-02472-z
- 7. Komorizono Y, Oketani M, Sako K, Yamasaki N, Shibatou T, Maeda M, et al. Risk factors for local recurrence of small hepatocellular carcinoma tumors after a single session, single application of percutaneous radiofrequency ablation. Cancer. 2003;97:1253-1262. https://doi.org/10.1002/cncr.11168
- 8. Teratani T, Yoshida H, Shiina S, Obi S, Sato S, Tateishi R, et al. Radiofrequency ablation for hepatocellular carcinoma in so-called high-risk locations. Hepatology. 2006;43:1101-1108. https://doi.org/10.1002/hep.21164
- 9. Han S, Sung PS, Park SY, Kim JW, Hong HP, Yoon JH, et al. Local ablation for hepatocellular carcinoma: 2024 expert consensus-based practical recommendation of the Korean Liver Cancer Association. J Liver Cancer. 2024;24:131-144. https://doi.org/10.17998/jlc.2024.08.04
- 10. Yang W, Yan K, Wu GX, Wu W, Fu Y, Lee JC, et al. Radiofrequency ablation of hepatocellular carcinoma in difficult locations: strategies and long-term outcomes. World J Gastroenterol. 2015;21:1554-1566. https://doi.org/10.3748/wjg.v21.i5.1554
- 11. Llovet JM, Vilana R, Bru C, Bianchi L, Salmeron JM, Boix L, et al. Increased risk of tumor seeding after percutaneous radiofrequency ablation for single hepatocellular carcinoma. Hepatology. 2001;33:1124-1129. https://doi.org/10.1053/jhep.2001.24233
- 12. Espinoza S, Briggs P, Duret JS, Lapeyre M, de Baere T. Radiofrequency ablation of needle tract seeding in hepatocellular carcinoma. J Vasc Interv Radiol. 2005;16:743-746. https://doi.org/10.1097/01.RVI.0000153109.56827.70
- 13. Jaskolka JD, Asch MR, Kachura JR, Ho CS, Ossip M, Wong F, et al. Needle tract seeding after radiofrequency ablation of hepatic tumors. J Vasc Interv Radiol. 2005;16:485-491. https://doi.org/10.1097/01.RVI.0000151141.09597.5F
- 14. Tanaka S, Shimada M, Shirabe K, Maehara S, Tsujita E, Taketomi A, et al. Surgical outcome of patients with hepatocellular carcinoma originating in the caudate lobe. Am J Surg. 2005;190:451-455. https://doi.org/10.1016/j.amjsurg.2004.12.005
- 15. Sakamoto Y, Nara S, Hata S, Yamamoto Y, Esaki M, Shimada K, et al. Prognosis of patients undergoing hepatectomy for solitary hepatocellular carcinoma originating in the caudate lobe. Surgery. 2011;150:959-967. https://doi.org/10.1016/j.surg.2011.03.005
- 16. Shimada S, Kamiyama T, Yokoo H, Orimo T, Nagatsu A, Ohata T, et al. Prognoses and clinicopathological characteristics for hepatocellular carcinoma originating from the caudate lobe after surgery. World J Surg. 2019;43:1085-1093. https://doi.org/10.1007/s00268-018-4869-2
- 17. Yamamoto T, Kubo S, Shuto T, Ichikawa T, Ogawa M, Hai S, et al. Surgical strategy for hepatocellular carcinoma originating in the caudate lobe. Surgery. 2004;135:595-603. https://doi.org/10.1016/j.surg.2003.10.015
- 18. Liu P, Yang JM, Niu WY, Kan T, Xie F, Li DQ, et al. Prognostic factors in the surgical treatment of caudate lobe hepatocellular carcinoma. World J Gastroenterol. 2010;16:1123-1128. https://doi.org/10.3748/wjg.v16.i9.1123
- 19. Seror O, Haddar D, N'Kontchou G, Ajavon Y, Trinchet JC, Beaugrand M, et al. Radiofrequency ablation for the treatment of liver tumors in the caudate lobe. J Vasc Interv Radiol. 2005;16:981-990. https://doi.org/10.1097/01.RVI.0000159859.71448.8A
- 20. Peng ZW, Liang HH, Chen MS, Zhang YJ, Li JQ, Zhang YQ, et al. Percutaneous radiofrequency ablation for the treatment of hepatocellular carcinoma in the caudate lobe. Eur J Surg Oncol. 2008;34:166-172. https://doi.org/10.1016/j.ejso.2007.08.004
- 21. Nishigaki Y, Tomita E, Hayashi H, Suzuki Y, Iritani S, Kato T, et al. Efficacy and safety of radiofrequency ablation for hepatocellular carcinoma in the caudate lobe of the liver. Hepatol Res. 2013;43:467-474. https://doi.org/10.1111/j.1872-034X.2012.01095.x
- 22. Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208-1236. https://doi.org/10.1002/hep.20933
- 23. Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68:723-750. https://doi.org/10.1002/hep.29913
- 24. Crocetti L, de Baere T, Lencioni R. Quality improvement guidelines for radiofrequency ablation of liver tumours. Cardiovasc Intervent Radiol. 2010;33:11-17. https://doi.org/10.1007/s00270-009-9736-y
- 25. Kang TW, Lim HK, Lee MW, Kim YS, Rhim H, Lee WJ, et al. Long-term therapeutic outcomes of radiofrequency ablation for subcapsular versus nonsubcapsular hepatocellular carcinoma: a propensity score matched study. Radiology. 2016;280:300-312. https://doi.org/10.1148/radiol.2016151243
- 26. Morimoto M, Numata K, Kondo M, Nozaki A, Hamaguchi S, Takebayashi S, et al. C-arm cone beam CT for hepatic tumor ablation under real-time 3D imaging. AJR Am J Roentgenol. 2010;194:W452-W454. https://doi.org/10.2214/AJR.09.3514
- 27. Lee MW, Kang D, Lim HK, Cho J, Sinn DH, Kang TW, et al. Updated 10-year outcomes of percutaneous radiofrequency ablation as first-line therapy for single hepatocellular carcinoma < 3 cm: emphasis on association of local tumor progression and overall survival. Eur Radiol. 2020;30:2391-2400. https://doi.org/10.1007/s00330-019-06575-0
- 28. European Association for the Study of the Liver. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. https://doi.org/10.1016/j.jhep.2018.03.019
- 29. Pietrobon V, Cesano A, Marincola F, Kather JN. Next generation imaging techniques to define immune topographies in solid tumors. Front Immunol. 2020;11:604967. https://doi.org/10.3389/fimmu.2020.604967
- 30. Lin CH, Ho CM, Wu CH, Liang PC, Wu YM, Hu RH, et al. Minimally invasive surgery versus radiofrequency ablation for single subcapsular hepatocellular carcinoma ≤ 2026-03-262 cm with compensated liver cirrhosis. Surg Endosc. 2020;34:5566-5573. https://doi.org/10.1007/s00464-019-07357-x
- 31. Luker GD. Imaging the immune tumor microenvironment to monitor and improve therapy. Radiology. 2021;298:133-134. https://doi.org/10.1148/radiol.2020203799
- 32. Zheng H, Liu K, Yang Y, Liu B, Zhao X, Chen Y, et al. Microwave ablation versus radiofrequency ablation for subcapsular hepatocellular carcinoma: a propensity score-matched study. Eur Radiol. 2022;32:4657-4666. https://doi.org/10.1007/s00330-022-08537-5
 , Chang Jin Yoon2,3,4
, Chang Jin Yoon2,3,4 , Chong-ho Lee1
, Chong-ho Lee1 , Kun Yung Kim1
, Kun Yung Kim1 , Eun Sun Jang4
, Eun Sun Jang4 , Jin-Wook Kim4
, Jin-Wook Kim4 , Seung Jae Lee5
, Seung Jae Lee5 , Sook-Hyang Jeong4
, Sook-Hyang Jeong4 , Minuk Kim6
, Minuk Kim6 , Jae Hwan Lee2,3,4,*
, Jae Hwan Lee2,3,4,*