Abstract
Acute SVC syndrome caused by extensive thrombosis requires prompt endovascular intervention. We report a 48-year-old female with colon cancer presenting with massive chemoport-related thrombosis involving the SVC, right atrium (RA), and bilateral brachiocephalic veins. Due to the lack of an embolic protection filter landing zone, we performed a novel plug-assisted thrombectomy (PAT) technique. A 20-mm vascular plug was positioned at the RA-SVC junction as a temporary tethered filter without detachment. Following mechanical thrombectomy, plug retrieval, and adjunctive balloon dilatation, the patient recovered and was discharged on day 12. This case demonstrates the technical feasibility of PAT as a proof-of-concept approach for embolic protection in patients with extensive SVC thrombosis where conventional filter placement is anatomically precluded.
-
Keywords: Superior vena cava syndrome; Thrombosis; Thrombectomy; Case reports
Introduction
Upper-extremity deep vein thrombosis (UEDVT) accounts for approximately 10% of all deep vein thrombosis (DVT) cases, and its incidence has been steadily increasing—a trend closely associated with the growing use of indwelling central venous catheters [
1,
2]. Subsequent thrombosis of the innominate and subclavian veins may occur with severe SVC stenosis. Nowadays, most non-malignant SVC syndromes are attributed to indwelling central venous catheters or pacemaker leads [
3]. The primary treatment for UEDVT is standard anticoagulation, aimed at alleviating symptoms, preventing thrombus progression, and reducing the risk of fatal pulmonary thromboembolism (PTE) [
4]. However, despite the rising clinical burden of catheter-associated UEDVT, there is a lack of established consensus or specific guidelines regarding its optimal management, particularly concerning anticoagulation protocols and the indications for SVC filter placement. When acute UEDVT progresses to massive SVC thrombosis, it can cause severe hemodynamic instability, necessitating endovascular intervention. While catheter-directed thrombolysis [
5,
6] or mechanical thrombectomy (e.g., the FlowTriever system) [
7] is increasingly utilized, supporting literature remains limited to case reports and small series. During aggressive mechanical thrombectomy for lower extremity DVT, an IVC filter is routinely and safely utilized to provide embolic protection [
8]. While some studies have reported on the safety of placing filters in the SVC to protect against PTE in patients with UEDVT, its routine use remains controversial [
9,
10]. In contrast, using a conventional filter in the SVC for UEDVT presents significant anatomical challenges due to the lack of an adequate landing zone, and carries catastrophic complication risks, such as caval perforation and cardiac tamponade [
11]. Herein, we report a case of fatal hemodynamic compromise caused by extensive acute SVC thrombosis. To overcome the anatomical constraints and risks associated with conventional filters, we successfully performed mechanical thrombectomy using a vascular plug as an embolic protection device (plug-assisted thrombectomy, PAT).
Case Report
A 48-year-old woman with a history of laparoscopic anterior resection for sigmoid colon cancer 2 years earlier and subsequent adjuvant chemotherapy presented with syncope and abrupt, severe swelling of the face and bilateral arms. She had an indwelling chemoport actively used for palliative chemotherapy due to recently developed lymph node and peritoneal metastases. Upon arrival, the patient was hemodynamically unstable with an initially unmeasurable blood pressure, which marginally stabilized to 97/80 mmHg with compensatory tachycardia (124 bpm) and respiratory alkalosis (pH 7.460, PaCO
2 13.5 mmHg, PaO
2 100.6 mmHg, and HCO
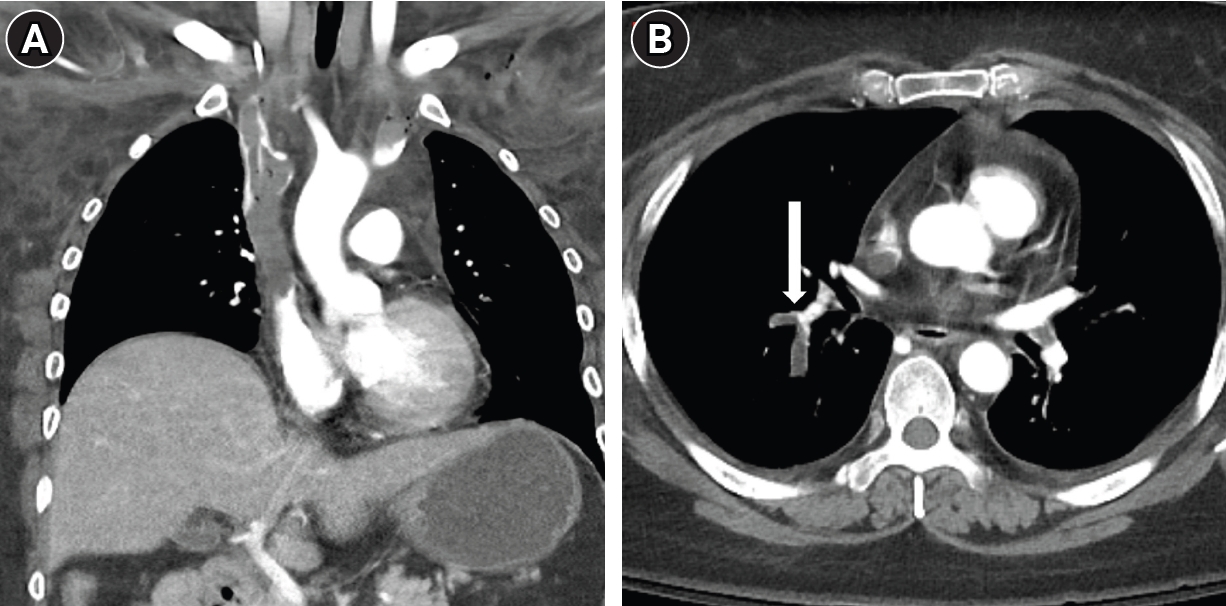
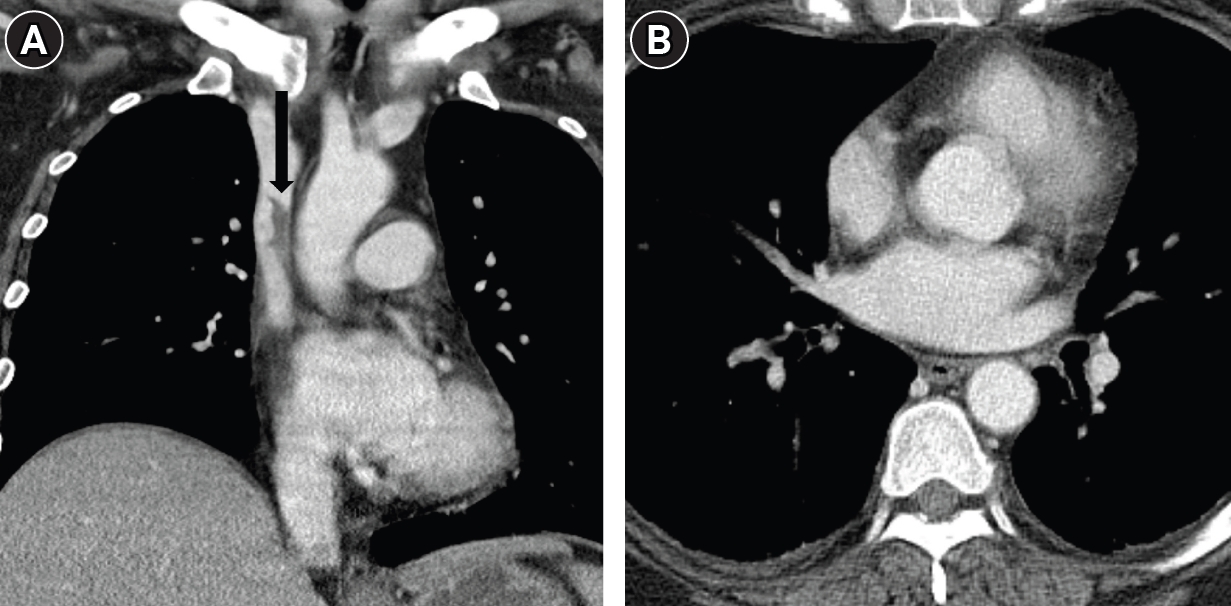
3 9.6 mmEq/L) on arterial blood gas analysis. Initial laboratory investigations revealed a significantly elevated D-dimer level, 30 µg/mL. Complete blood count showed anemia, with a hemoglobin of 8.8 g/dL and a hematocrit of 27.1%, while the platelet count was 141 × 10³/μL. Coagulation studies indicated an INR of 1.62, with a corresponding prothrombin time of 18.5 seconds. Contrast-enhanced chest CT revealed extensive, diffuse thrombosis involving the bilateral subclavian, brachiocephalic veins, and the SVC, extending into the right atrium (RA) (
Fig. 1A). Acute PTE was identified in right lower lobar and segmental pulmonary arteries (
Fig. 1B). Given the massive thrombus burden, urgent endovascular thrombectomy was planned. At the time of intervention, the patient required dual vasopressor support with norepinephrine (0.24 µg/kg/min) and vasopressin (0.02 units/min), maintaining a blood pressure of 84/59 mmHg. To evaluate the risk of acute right ventricular (RV) volume overload following restoration of SVC flow, preprocedural echocardiography was performed and demonstrated no evidence of RV strain or dysfunction. Although extracorporeal membrane oxygenation was considered as a rescue strategy, it was deferred after multidisciplinary discussion given the patient’s underlying malignancy and estimated prognosis of less than one year. Due to the severe risk of massive PTE during the procedure, prophylactic endotracheal intubation was performed under general anesthesia. When planning the procedure, our primary concern was how to effectively prevent thrombus migration into the RA during thrombectomy. We initially considered the use of an IVC filter-type device as a temporary barrier; however, given the absence of an appropriate landing zone within the SVC, this approach was not feasible. Instead, we considered a vascular plug, which offered both a sufficiently large diameter to be deployed within the SVC and the mechanical properties necessary to function as a temporary barrier against thrombus fragment migration during thrombectomy.
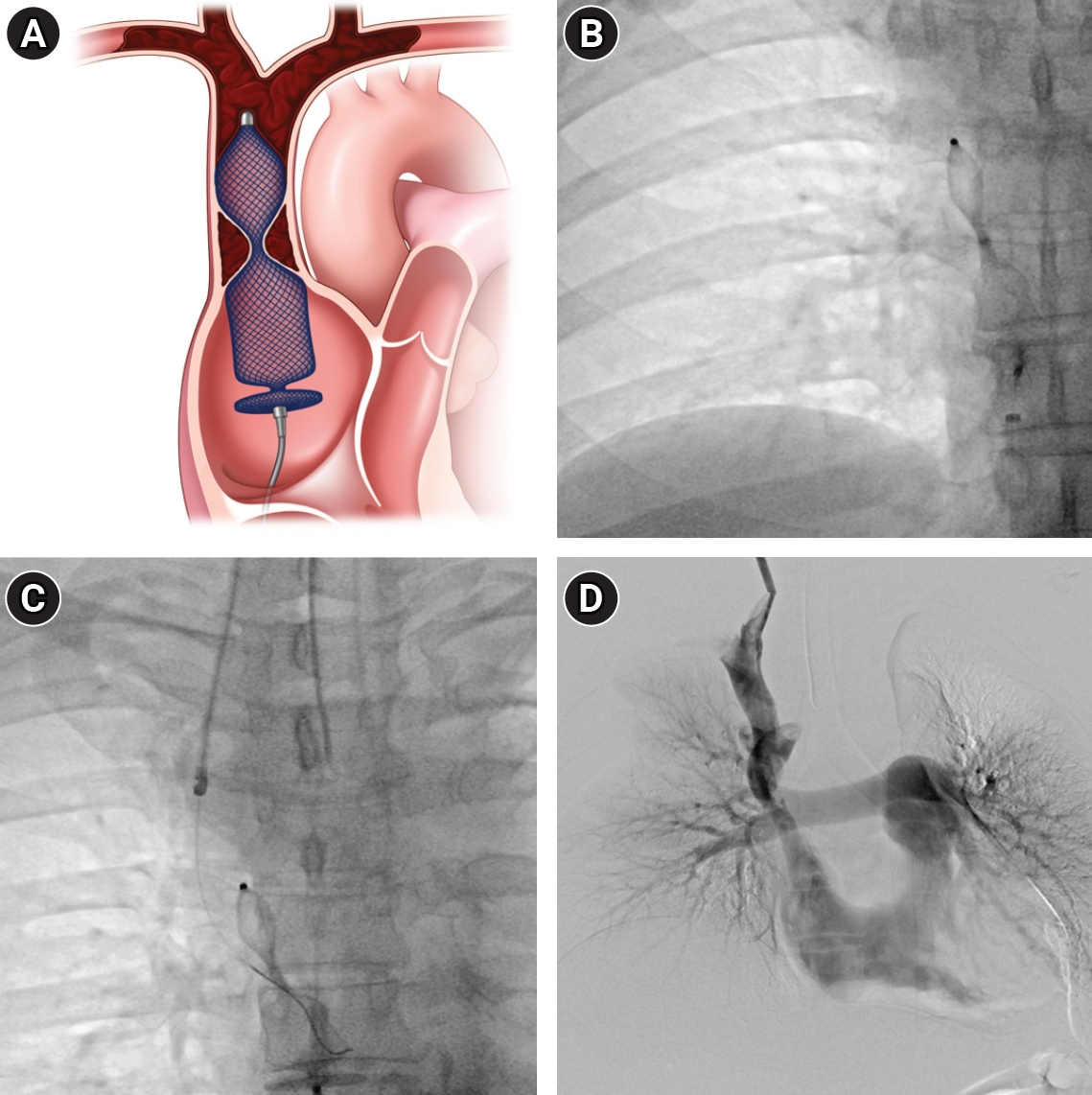
Right common femoral vein access was achieved under ultrasound guidance using a micropuncture set with a 21-gauge needle. A 7-F Shuttle Flexor guiding sheath (Cook Inc., Bloomington, IN, USA) was then placed, through which a 5-F C2 Cobra catheter (Boston Scientific, Cork, Ireland) was advanced. Venography through the 5-F angiographic catheter in the RA showed thrombotic occlusion and narrowing at the SVC-RA junction. Since the diameter of the RA-SVC junction was measured at 13 mm on venography, plug sizing was initially attempted with a 22-mm Amplatzer Vascular Plug II (Abbott Medical, Abbott Park, IL, USA); however, this plug failed to maintain stable positioning and was dislodged, likely due to excessive oversizing. A 20-mm plug was subsequently deployed, which provided adequate and stable anchoring throughout the procedure. To prevent potential migration, one lobe of the plug was intentionally positioned within the thrombotic segment, ensuring stable anchoring and complete occlusion at the junction. Crucially, instead of detachment, the plug was only deployed and maintained on the delivery cable throughout the procedure, serving as a temporary sieve and mechanical barrier against large emboli (
Fig. 2A,
2B). A second access was established via the right internal jugular vein using ultrasound guidance and a micropuncture set. Initially, mechanical thrombectomy using an 8-F Aspirex device (Straub Medical AG, Wangs, Switzerland) was performed to reduce the overall thrombotic burden. During this process, the use of the Aspirex device was strictly avoided in the vicinity of the vascular plug to prevent potential device damage or entanglement (
Fig. 2C). During plug retrieval, a critical technical consideration was the directionality of the capture maneuver. Rather than advancing the sheath toward the plug—which risks mechanically displacing the surrounding thrombus caudally into the RA—the 7-F Shuttle Flexor sheath was maintained in a stationary position while the plug was retracted cranially by pulling the delivery cable, drawing the plug back into the fixed sheath. This ‘device-to-sheath’ retrieval technique minimizes mechanical interaction with the peri-plug thrombus and reduces the risk of iatrogenic embolization during retrieval. Following the retrieval of the plug, residual thrombosis adjacent to the plug site was safely removed via manual aspiration using a 7-F Envoy guiding catheter (Codman Neurovascular, Raynham, MA, USA). Following the aspiration of the massive thrombus, a residual stenosis was identified in the SVC adjacent to the RA. Balloon angioplasty was performed using a 12 × 60-mm Mustang balloon (Boston Scientific, Marlborough, MA, USA). Final venography revealed that the main flow of the SVC was restored (
Fig. 2D). Residual thrombus in the SVC and bilateral brachiocephalic veins was planned to be managed with anticoagulation. Following the procedure, anticoagulation was administered according to our institutional standard-dose heparinization protocol: an initial bolus of unfractionated heparin 3,000 IU was administered intravenously, followed by a continuous infusion of 20,000 IU in 500 mL normal saline at 20 mL/h (800 IU/h), targeting an aPTT of 50–75 seconds.
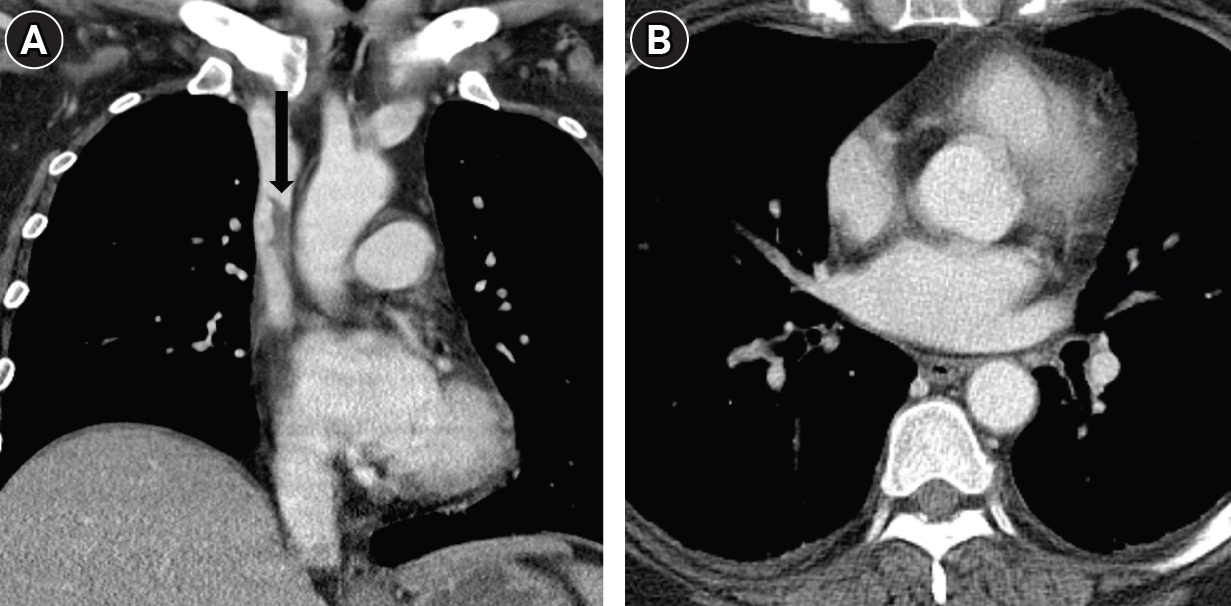
The patient showed dramatic clinical improvement, with edema resolving over the next 10 days. A follow-up CT demonstrated near-complete resolution of the thrombosis (
Fig. 3). She was successfully extubated and discharged 12 days after the initial presentation. At discharge, the patient was maintained on prophylactic-dose enoxaparin (40 mg once daily), as therapeutic-dose anticoagulation was de-escalated due to the development of hematuria during admission. Outpatient follow-up was scheduled with repeat laboratory evaluation including D-dimer.
Discussion
The management of extensive SVC thrombosis involving hemodynamic instability and a high thrombus burden extending into the RA remains clinically challenging. In the present case, the patient’s clinical deterioration—characterized by syncope, hypotension, and severe respiratory alkalosis—necessitated immediate endovascular intervention. However, the existing PTE and the massive thrombus burden at the SVC-RA junction presented a significant procedural dilemma: the high risk of treatment-induced fatal pulmonary embolism versus the lack of a safe landing zone for conventional embolic protection.
The anatomical constraints of the SVC often preclude the use of SVC filters, which are associated with risks of migration, caval perforation, and cardiac tamponade in this shorter and more mobile vessel segment [
9,
11]. Furthermore, since the thrombus in our patient had already extended adjacent to the RA, there was insufficient space for safe filter deployment. To overcome these challenges, we utilized a vascular plug as a temporary embolic protection device. We hypothesized that the fine nitinol mesh of the Amplatzer Vascular Plug II could similarly serve as a mechanical barrier against large, procedurally-dislodged thrombi.
The use of a vascular plug as a protection device in the SVC offers several distinct clinical advantages. First, when the plug is not detached from the delivery cable, it can be repositioned freely to accommodate real-time changes in thrombus morphology and can be easily retrieved into the sheath once the thrombectomy is completed [
12]. Second, the availability of various sizes (2–22 mm) allows for precise fitting in large-caliber vessels like the SVC, ensuring a stable "sieve" effect. Third, the safety profile of these plugs is well-established across various clinical scenarios with minimal complications [
13]. Although a delivery sheath of 7 F or larger is required, this is rarely a limiting factor in the context of large-bore SVC interventions. However, one important caveat is that plug stability may diminish as thrombectomy progresses. As the surrounding thrombus burden is reduced, the thrombus that initially contributes to plug anchoring is gradually eliminated, potentially rendering the plug susceptible to dislodgement. Continuous fluoroscopic monitoring of plug position throughout the procedure is therefore strongly advisable.
In this case, diffuse thrombosis in the SVC and right brachiocephalic vein was successfully resolved. The clinical improvement and the patient’s discharge within 12 days underscore the feasibility of this technique. For patients with massive SVC thrombosis where the risk of procedural PTE is high and conventional filter placement is anatomically constrained, the use of a vascular plug as a temporary embolic protection device (PAT) may offer a feasible and versatile technical alternative.
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
None.
Acknowledgments
None.
Author contributions
.
Data availability statement
.
Fig. 1.A 48-year-old woman with a history of sigmoid colon cancer presented with syncope and abrupt, severe swelling of the face and bilateral arms. (A) CT image shows extensive, diffuse thrombosis involving the SVC. (B) Axial CT image at the level of the lower thorax reveals acute pulmonary thromboembolism in the right lower lobar segmental pulmonary arteries (arrow).
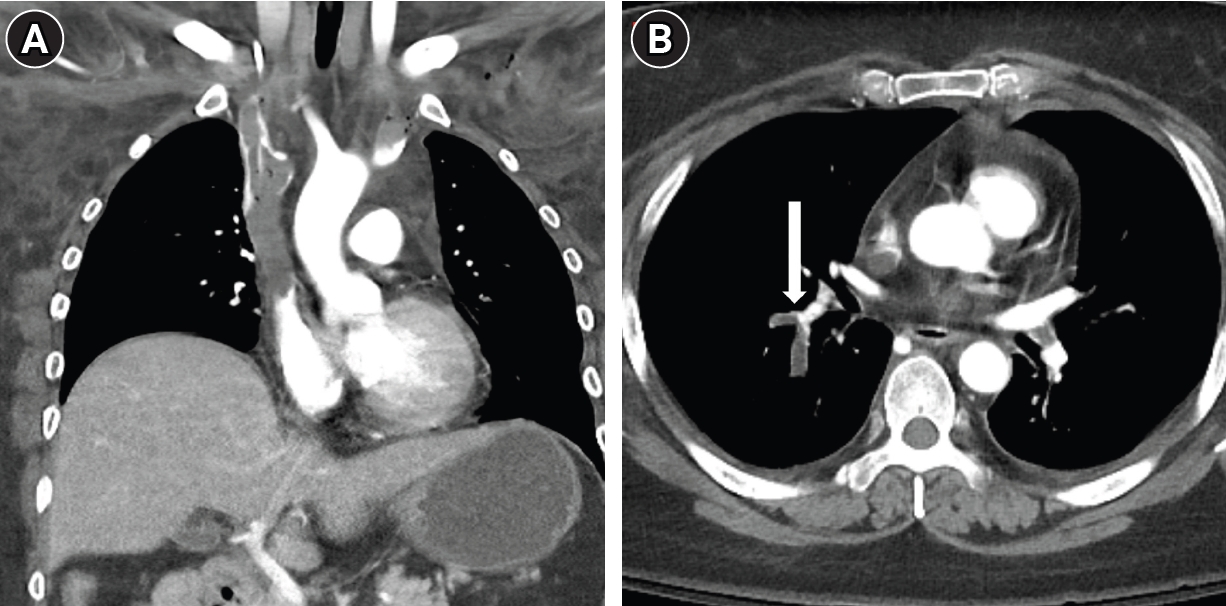
Fig. 2.Endovascular thrombectomy. (A) Illustration of a vascular plug deployed at the SVC–right atrium (RA) junction to prevent the migration of massive thrombus. (B) Vascular plug is deployed at the junction of SVC-RA, where the portion of vascular plug in the thrombosed SVC is partially opened. (C) Mechanical thrombectomy is performed via route of right internal jugular vein. (D) The deployed vascular plug was safely retrieved, with a small amount of residual thrombus remaining in the SVC.
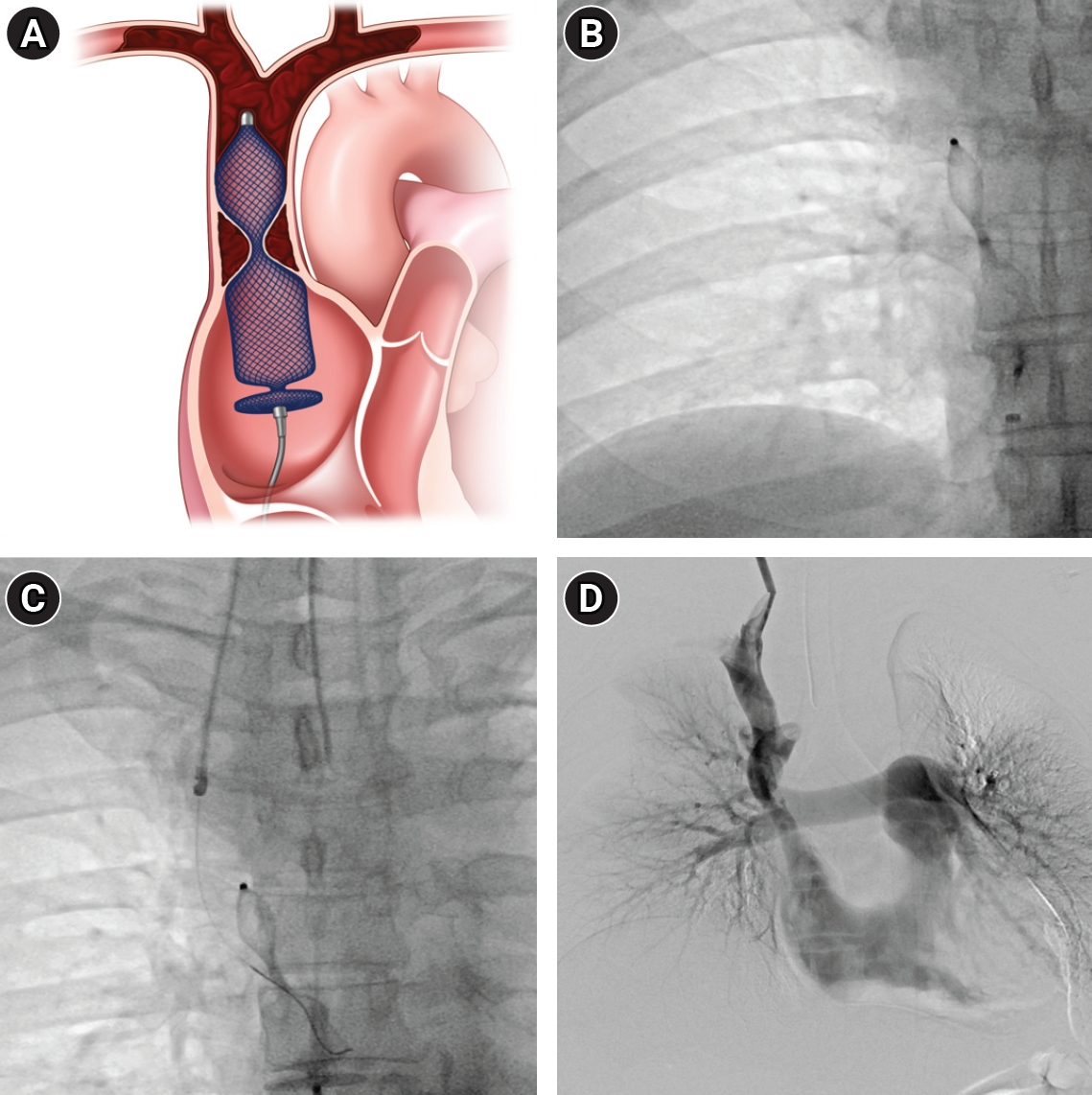
Fig. 3.Follow-up. (A) A 10-day follow-up CT scan shows near-complete resolution of the thrombus, with a small amount of residual thrombus remaining in the superior vena cava (arrow). (B) Axial CT image reveals complete resolution of the previous pulmonary thromboembolism, with no evidence of newly developed PTE.
References
- 1. Munoz FJ, Mismetti P, Poggio R, Valle R, Barron M, Guil M, et al. Clinical outcome of patients with upper-extremity deep vein thrombosis: results from the RIETE Registry. Chest. 2008;133:143-148. https://doi.org/10.1378/chest.07-1432
- 2. Joffe HV, Kucher N, Tapson VF, Goldhaber SZ; Deep Vein Thrombosis (DVT) FREE Steering Committee. Upper-extremity deep vein thrombosis: a prospective registry of 592 patients. Circulation. 2004;110:1605-1611. https://doi.org/10.1161/01.CIR.0000142289.94369.D7
- 3. Rice TW, Rodriguez RM, Light RW. The superior vena cava syndrome: clinical characteristics and evolving etiology. Medicine (Baltimore). 2006;85:37-42. https://doi.org/10.1097/01.md.0000198474.99876.f0
- 4. Engelberger RP, Kucher N. Management of deep vein thrombosis of the upper extremity. Circulation. 2012;126:768-773. https://doi.org/10.1161/CIRCULATIONAHA.111.051276
- 5. Cui J, Kawai T, Irani Z. Catheter-directed thrombolysis in acute superior vena cava syndrome caused by central venous catheters. Semin Dial. 2015;28:548-551. https://doi.org/10.1111/sdi.12362
- 6. Carroll H, Dineen F, Ryan P, Crowley M, Power S, O'Reilly S. Catheter-directed thrombolysis for acute SVC obstruction in a patient with metastatic breast cancer: catheter-directed thrombolysis for SVC obstruction. Arch Breast Cancer. 2022;9:407-410. https://doi.org/10.32768/abc.202293407-410
- 7. Sousou JM, Sherard DM, Edwards JR, Negron-Rubio E. Successful removal of a thrombus in the setting of SVC syndrome using the INARI FlowTriever device. Radiol Case Rep. 2022;17:744-747. https://doi.org/10.1016/j.radcr.2021.12.032
- 8. Lee SH, Kim HK, Hwang JK, Kim SD, Park SC, Kim JI, et al. Efficacy of retrievable inferior vena cava filter placement in the prevention of pulmonary embolism during catheter-directed thrombectomy for proximal lower-extremity deep vein thrombosis. Ann Vasc Surg. 2016;33:181-186. https://doi.org/10.1016/j.avsg.2015.10.034
- 9. Usoh F, Hingorani A, Ascher E, Shiferson A, Tran V, Marks N, et al. Long-term follow-up for superior vena cava filter placement. Ann Vasc Surg. 2009;23:350-354. https://doi.org/10.1016/j.avsg.2008.08.012
- 10. Yim NY, Chang NK, Lim JH, Kim JK. Retrograde Tempofilter II placement within the superior vena cava in a patient with acute upper extremity deep venous thrombosis: the filter stands on its head. Korean J Radiol. 2011;12:140-143. https://doi.org/10.3348/kjr.2011.12.1.140
- 11. Hussain SM, McLafferty RB, Schmittling ZC, Zakaria AM, Ramsey DE, Larson JL, et al. Superior vena cava perforation and cardiac tamponade after filter placement in the superior vena cava: a case report. Vasc Endovascular Surg. 2005;39:367-370. https://doi.org/10.1177/153857440503900412
- 12. Vaidya S, Tozer KR, Chen J. An overview of embolic agents. Semin Intervent Radiol. 2008;25:204-215. https://doi.org/10.1055/s-0028-1085930
- 13. Lopera JE. The Amplatzer vascular plug: review of evolution and current applications. Semin Intervent Radiol. 2015;32:356-369. https://doi.org/10.1055/s-0035-1564810
Citations
Citations to this article as recorded by