Abstract
Pyrrolizidine alkaloid–induced hepatic sinusoidal obstruction syndrome (PA-HSOS) is highly prevalent in Asia, primarily due to the widespread use of traditional herbal medicines containing hepatotoxic pyrrolizidine alkaloids. This condition poses significant clinical challenges, including diagnostic difficulties and limited therapeutic options, frequently leading to severe liver damage and portal hypertension. Transjugular intrahepatic portosystemic shunt (TIPS) treatment has emerged as a key intervention for managing complications associated with PA-HSOS, such as refractory ascites and variceal bleeding, by reducing portal pressure and supporting liver function recovery. However, TIPS has not been widely accepted as a salvage therapy for severe PA-HSOS unresponsive to anticoagulation therapy, mainly due to concerns about post-TIPS complications, particularly hepatic encephalopathy. Consequently, careful patient selection and risk stratification are critical. This review synthesizes the current evidence on PA-HSOS in Asia, evaluates the clinical utility of TIPS, and discusses strategies to optimize outcomes while minimizing adverse effects. Specifically, we review the epidemiology, pathophysiology, and diagnostic advancement of PA-HSOS, with a particular focus on the evolving role of TIPS in its management.
-
Keywords: Hepatic sinusoidal obstruction syndrome; Pyrrolizidine alkaloids; Portal hypertension; Transjugular intrahepatic portosystemic shunt; Treatment
Introduction
Hepatic sinusoidal obstruction syndrome (HSOS) is a vascular liver disorder characterized by damage to hepatic sinusoidal endothelial cells, resulting in sinusoidal congestion, portal hypertension (PH), and potential liver failure [
1-
3]. In Western countries, HSOS is predominantly associated with hematopoietic stem cell transplantation (HSCT) or certain chemotherapeutic agents [
4,
5]. On the other hand, pyrrolizidine alkaloid (PA)–induced HSOS (PA-HSOS) in Asia is primarily induced by the consumption of traditional herbal medicines containing PAs, such as
Gynura japonica [
1,
6]. There are notable clinical and histopathological differences between the two entities. Clinically, PA-HSOS typically presents with abdominal pain, ascites, jaundice, and liver dysfunction, whereas HSCT-related HSOS is more frequently associated with rapid weight gain and renal impairment [
7]. Histopathologically, PA-HSOS is characterized by prominent sinusoidal congestion and hepatocyte necrosis, whereas HSCT-related HSOS lacks these specific damage mechanisms inherent to PA exposure [
8]. Despite growing clinical recognition, PA-HSOS remains a diagnostic challenge due to overlapping features with other causes of acute liver injury and PH [
9]. Histopathologically, PA-HSOS is characterized by sinusoidal dilation, erythrocyte extravasation, and fibrosis, which can be confirmed by liver biopsy [
10,
11].
PA-HSOS constitutes a substantial health burden in several Asian countries, particularly China, South Korea, and India, where the use of traditional herbal medicines is widespread [
12-
15]. The market size and diversity of herbal products vary across these countries [
16,
17]. South Korea shows a clear preference for traditional herbal products, particularly with a growing trend in dietary supplements. A survey of 1,134 Korean respondents revealed that 726 individuals had used herbal products, indicating a high prevalence rate [
16]. Notably, Socheongryongtang is the most commonly used herbal formula for treating allergic rhinitis [
18]. Similarly, India, often referred to as the 'World's Medicinal Plant Garden,' possesses approximately 8,000 medicinal plant species and provides an estimated 25,000 effective herbal formulations [
19,
20]. In China, PA-HSOS accounts for a substantial proportion (50.0%–88.6%) of all HSOS cases, frequently linked to herbal products like
Gynura japonica [
21]. Retrospective studies from China have identified PA-HSOS as a leading cause of drug-induced liver injury (DILI) [
11,
22]. The condition is closely associated with unregulated herbal products, which underscores the need for greater public awareness and regulatory control [
10,
23]. Socioeconomic factors, including reliance on folk remedies in rural populations, further increase the risk of PA-HSOS [
24]. The condition predominantly affects older individuals and those with chronic exposure to herbal substances, emphasizing the need for targeted preventive strategies [
10,
25].
Clinical management of PA-HSOS differs notably from Western HSOS, reflecting distinct etiologies and pathophysiology. In PA-HSOS, early anticoagulation or transjugular intrahepatic portosystemic shunt (TIPS) is commonly pursued, especially in severe cases, to reduce portal pressure and improve hepatic perfusion. Liver transplantation is considered for refractory PA-HSOS. By contrast, Western guidelines emphasize defibrotide prophylaxis and broader supportive care as central strategies, with liver transplantation reserved for refractory cases. TIPS appears to show superior outcomes over conservative therapy specifically in PA-HSOS but is rarely used and less favored in Western HSOS. These differences underscore the need for region-specific, tailored management algorithms that account for exposure patterns, resource availability, and clinical trajectories.
This review highlights the key clinical challenges of PA-HSOS, including delayed diagnosis due to nonspecific manifestations such as ascites and hepatomegaly, as well as the lack of reliable biomarkers. We evaluate the current evidence supporting the use of TIPS as a therapeutic option, noting its efficacy in reducing portal pressure despite variable post-procedural outcomes. Management strategies for PA-HSOS are compared with Western HSOS protocols, with a focus on region-specific considerations in Asia. The review also explores emerging treatments, including anticoagulation therapy (AT) with rivaroxaban and gut microbiota modulation. Finally, we advocate for the adoption of standardized diagnostic frameworks, such as the Drum Tower Severity Scoring (DTSS) system, to improve diagnostic accuracy and consistency in PA-HSOS management.
Epidemiology and Etiology
Epidemiology
PA-HSOS is a significant public health concern in Asia, primarily attributed to the widespread use of herbal medicines and dietary supplements containing PAs. In China, PA-HSOS has emerged as the leading cause of DILI, with PA-rich herb products, such as
Gynura japonica, playing a central role in its etiology [
26]. A systematic review based on 2,156 reported cases of HSOS related to
Gynura japonica from 1980 to 2019 indicated that HSOS caused by this herb accounts for over 50% of DILI cases [
26]. The cultural reliance on traditional herbal remedies, often consumed without adequate awareness of their hepatotoxic potential, further contributes to the high prevalence of PA-HSOS (
Table 1). However, despite its clinical relevance, comprehensive epidemiological data on PA-HSOS remain limited, as the majority of existing studies are retrospective and involve small cohorts. This highlights a critical gap in systematic surveillance and standardized reporting in the region [
1,
27].
The hepatotoxicity of PAs is primarily mediated by metabolic conversion into reactive intermediates, such as dehydro-PAs, which directly damage liver sinusoidal endothelial cells (LSECs) [
26,
28]. This injury disrupts the sinusoidal architecture, leading to the characteristic features of HSOS, including hepatomegaly, hyperbilirubinemia, and ascites [
3,
26]. LSECs are particularly vulnerable to PA-induced damage due to their fenestrated structure and high exposure to circulating toxins. The resulting injury to LSECs triggers a cascade of pathological events, including sinusoidal capillarization, perisinusoidal matrix deposition, and impaired vascular exchange, which are key hallmarks of HSOS [
29,
30]. Additionally, PAs are known to disrupt bile acid homeostasis, aggravating liver injury. The imbalance in bile acid metabolism leads to heightened oxidative stress and hepatocellular damage, contributing to the progression of HSOS [
31,
32].
Emerging evidence further implicates gut microbiota dysbiosis in PA-HSOS pathogenesis. Experimental models have shown that fecal microbiota transplantation can modulate disease severity, suggesting a role for the gut-liver axis in PA-HSOS development [
33,
34]. Additionally, macrophage activation and subsequent inflammatory responses amplify sinusoidal injury, creating a pro-fibrogenic microenvironment [
29,
34]. Over time, PA-HSOS may progress to chronic liver disease, with the development of hepatic fibrosis; however, the precise mechanisms underlying this transition remain incompletely understood [
30,
35].
Middle-aged and elderly individuals with prolonged exposure to PA-containing herbal products are the highest-risk group for developing PA-HSOS [
27,
36]. The early mortality rate in this population is approximately 10.3% to 14%, and the 3-year cumulative mortality ranges from 24% to 30.8% [
37,
38]. Notably, in patients receiving only supportive care, mortality can reach as high as 43.9% [
39]. This high mortality rate is concerning, as these individuals often rely on traditional remedies for chronic conditions, typically without awareness of their potential hepatotoxic effects. For instance,
Gynura japonica, a popular herb used for treating blood stasis or traumatic injuries, has been strongly associated with numerous cases of HSOS due to its high PAs content [
36]. Inadequate regulation of herbal products in many Asian countries exacerbates the risk, as contamination or misidentification of PA-producing plants is not uncommon [
32,
40]. Reducing the burden of PA-HSOS in these populations requires a multifaceted approach, including enhanced public education, stricter regulatory oversight, and improved strategies for early diagnosis. Additionally, the effects of PAs on individuals are stochastic, characterized by significant variability [
41]. Evidence indicates that individual differences in metabolism, species sensitivity, and chemical structure lead to diverse toxic responses, underscoring the non-deterministic nature of these effects rather than a uniform response across all individuals [
42].
Diagnosis
The diagnosis of PA-HSOS relies on a comprehensive assessment that includes clinical features, imaging findings, and histopathological evaluation [
15]. Patients typically present with the hallmark triad of abdominal distension, hepatic region pain, and ascites, often accompanied by jaundice and hepatomegaly (
Fig. 1). The widely adopted "Nanjing criteria" for PA-HSOS diagnosis require a confirmed history of exposure to PA-containing plants, such as
Gynura segetum, along with at least one of the following: abdominal or hepatic symptoms, elevated serum bilirubin or abnormal liver function tests, imaging evidence of hepatic vascular changes, or compatible pathological findings after excluding other liver injuries [
15].
In addition to these criteria, the validated DTSS system has proven useful in stratifying disease severity and guiding therapeutic decisions [
27,
43]. Imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI) play an important non-invasive role in the diagnostic process, often revealing characteristic signs such as hepatic sinusoidal congestion, hepatic vein narrowing, and a "clover-like" enhancement pattern [
3,
44] (
Fig. 1). Enhanced CT typically shows heterogeneous low-density areas in the liver, which is the most common manifestation, alongside hepatomegaly, ascites, and thickening of the gallbladder wall. During the arterial or portal venous phase, patchy liver enhancement reflects abnormal perfusion [
45]. On MRI, gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid (Gd-EOB-DTPA) enhanced imaging during the hepatobiliary phase consistently shows low signal intensity in PA-HSOS patients, correlating positively with prothrombin time (PT) and international normalized ratio, thus serving as an independent mortality risk factor. Additionally, susceptibility-weighted imaging and T2-weighted imaging reveal low signal areas associated with hemosiderin deposition [
46]. Nevertheless, liver biopsy remains the diagnostic gold standard, allowing for the identification of characteristic histopathological lesions. Differentiating PA-HSOS from other vascular liver diseases, including Budd-Chiari syndrome and cirrhotic PH, continues to present a significant clinical challenge (
Table 2) [
47,
48]. Future research should prioritize the validation of non-invasive biomarkers and the refinement of scoring systems such as DTSS to enhance diagnostic accuracy and allow timely intervention, particularly in high-prevalence regions.
Treatment Strategies
Medical Management
The treatment of PA-HSOS remains challenging due to the lack of specific therapeutic options. AT, including low-molecular-weight heparin (LMWH) and rivaroxaban, is often employed as an initial treatment. Specifically, LMWH is dosed at 4,000 IU subcutaneously twice daily [
39]. In a study involving five PA-HSOS patients treated with LMWH for 8–21 days, ascites resolved, symptoms improved, and hepatic venous blood flow was restored [
49]. However, in severe cases, AT alone appears limited in efficacy. Another study reported a mortality rate of 34.1% with AT versus 0% with TIPS [
50]. A multicenter study involving 249 PA-HSOS patients indicated that AT improved survival in some cases; however, the benefits were inconsistent, highlighting the need for better patient stratification [
39]. Defibrotide, which has proven effective for managing HSOS after HSCT, has also been studied in PA-HSOS [
51]. In HSCT-associated HSOS, defibrotide is dosed at 6.25 mg/kg intravenously every 6 hours (equivalent to 25 mg/kg per day) and continued for at least 3 weeks until total bilirubin (TBIL) levels normalize. However, its efficacy may be constrained by the unique pathogenesis of PA-HSOS, and its limited availability in China raises uncertainties about its effectiveness in treating PA-induced HSOS. A study assessing defibrotide effectiveness in monocrotaline-induced rat HSOS found that it improved outcomes, suggesting that defibrotide may be a preferable option to LMWH in clinical practice [
8].
TIPS has emerged as an important therapeutic option for PA-HSOS patients with refractory PH or ascites, and although it has no specific contraindications for treatment, guidelines recommend considering it for patients with ineffective medical therapy, highlighting the importance of individualized assessment [
15]. Studies have shown that ascites and pleural effusions can markedly improve within a short period after TIPS placement, with previously obstructed hepatic veins often regaining patency in the short term when combined with AT (
Figs. 1,
2) [
10,
52]. Various studies have confirmed the safety and efficacy of TIPS in managing PA-HSOS (
Table 3) [
50,
52-
57]. A retrospective study involving 30 PA-HSOS patients demonstrated that TIPS significantly reduced portal pressure, alleviated ascites, and improved clinical symptoms [
52]. The median time to complete ascites remission was 52 days, while the median recovery time for liver CT radiological manifestations was 196.5 days. However, the timing of the procedure is critical, with preoperative TBIL levels identified as a key prognostic factor. Early intervention, when TBIL levels are below 10 mg/dL, is associated with improved recovery [
53].
In the context of treatment strategy selection, a multicenter study comparing the efficacy between TIPS and AT in 164 PA-HSOS patients revealed that TIPS conferred significantly better mid- to long-term survival rates compared with those of AT, particularly in patients with moderate to severe PA-HSOS [
54]. This suggests that TIPS should be considered a viable initial treatment option for this patient cohort [
50]. Similarly, another retrospective study involving 69 patients in the TIPS group and 95 patients in the supportive treatment group confirmed that TIPS placement significantly improved survival rates and effectively alleviated PH-related clinical symptoms in PA-HSOS patients [
54].
Despite its promising efficacy, the use of TIPS in Asian populations faces several challenges. Research indicates that a significant proportion of PA-HSOS patients present at advanced stages of the disease, complicating the identification of appropriate candidates for TIPS [
58]. Additionally, postoperative anticoagulation management protocols vary widely among medical institutions, and there is currently no standardized approach for preventing shunt thrombosis or disease progression [
59]. To address these challenges, a research team developed the DTSS system, which incorporates prognostic factors such as liver function and imaging findings to guide treatment strategy selection, including TIPS candidacy [
11,
27].
Regarding prognostic assessment, several studies have explored predictive factors that may influence the effectiveness of TIPS treatment. One study found that prolonged baseline PT and elevated serum TBIL levels 5 days post-TIPS are independent risk factors predicting mortality in PA-HSOS patients following TIPS placement [
55]. Another study suggested that preoperative measurement of the hepatic venous pressure gradient may assist in assessing disease severity and predicting TIPS treatment efficacy [
60]. Notably, the response to TIPS may vary depending on the underlying cause of PA-HSOS. For example, a study involving nine patients with HSOS related to
Gynura segetum found that TIPS treatment significantly improved clinical symptoms; nevertheless, further research is needed to determine the optimal timing for intervention [
61]. Additionally, pathological studies have shown that TIPS placement can lead to significant improvements in pathological features, such as hepatic congestion and hepatocyte swelling, over time [
56].
Despite the promising outcomes of TIPS, its long-term efficacy in PA-HSOS requires further validation. A multicenter study involving 117 PA-HSOS patients emphasized the importance of establishing a reliable prognostic assessment system to better guide treatment decisions [
38]. Additionally, for some advanced PA-HSOS patients, TIPS may serve primarily as a bridge to liver transplantation [
25,
62]. In PA-HSOS patients without severe underlying cirrhosis, post-TIPS complications, such as hepatic encephalopathy (HE), are less common than in cirrhotic cohorts [
52,
63]. A systematic review included 19 studies and 465 HSOS patients, reported an overall HE incidence of 13.2% post-TIPS; however, this data encompasses all types of HSOS and is not limited to the PA-HSOS subgroup [
64]. Conversely, a retrospective review of 30 PA-HSOS patients who underwent TIPS found no post-procedure HE cases [
52]. By comparison, the pooled HE incidence among cirrhotic patients ranges from 33.2% to 58% [
65,
66]. The absence of advanced liver disease likely reduces the risks associated with portosystemic shunting, including hyperammonemia and HE, making TIPS a relatively safer option for these patients [
10,
25]. Overall, existing evidence suggests that TIPS may be an effective means of managing PA-HSOS associated with PH; however, careful consideration of patient-specific factors is essential to determine the appropriate indications and timing for intervention. Future research should focus on establishing standardized preoperative assessment systems and postoperative management protocols to further enhance treatment outcomes [
7,
45,
67].
For end-stage PA-HSOS patients who do not respond to medical and TIPS therapies, liver transplantation is the last resort. However, data on transplantation outcomes in PA-HSOS are limited, as most cases are managed conservatively or with TIPS [
25]. Although the high mortality rate and rapid disease progression highlight the importance of early referral to transplant centers, organ availability and patient selection criteria complicate the transplantation process, particularly in Asia [
38]. Future studies should aim to optimize the timing of liver transplantation and refine post-transplant management strategies to improve survival outcomes in this critically ill population.
Conclusion
Recent studies have advanced our understanding of PA-HSOS, particularly regarding the underlying toxicity pathways and the role of the gut-liver axis in disease progression. Emerging evidence suggests that the PU.1 signaling pathway plays a vital role in mediating PA-induced hepatotoxicity, primarily by driving damage to sinusoidal endothelial cells [
68]. Additionally, the gut microbiota has been implicated in PA-HSOS pathogenesis, with studies on fecal microbiota transplantation revealing that dysbiosis exacerbates liver injury through macrophage activation [
33,
34]. Experimental models of monocrotaline-induced HSOS have demonstrated the potential therapeutic benefits of modulating the gut microbiota [
34,
69], further underscoring the importance of investigating specific microbial taxa and their interactions with host immune pathways.
The lack of standardized diagnostic criteria for PA-HSOS in Asia highlights the urgent need for multicenter collaborative efforts to establish region-specific guidelines. Current diagnostic challenges include the overlap of PA-HSOS with other liver diseases and the variable sensitivity of non-invasive diagnostic tools [
70]. Furthermore, although TIPS is crucial for managing severe PA-HSOS, its clinical efficacy is often hampered by complications and inconsistent outcomes [
55]. The development of enhanced TIPS techniques and public education on the risks associated with the unregulated use of herbal medicines are essential for prevention and improved patient care [
71].
Conflict of interest
No potential conflicts of interest relevant to this article were reported.
Funding
None.
Acknowledgments
None.
Author contributions
Conceptualization, investigation, and manuscript writing: TYZ. Conceptualization and investigation: HLW, ZCJ. All aspects of the work: BX, JHS.
Data availability statement
Data sharing does not apply to this article as no datasets were generated or analyzed during the current study.
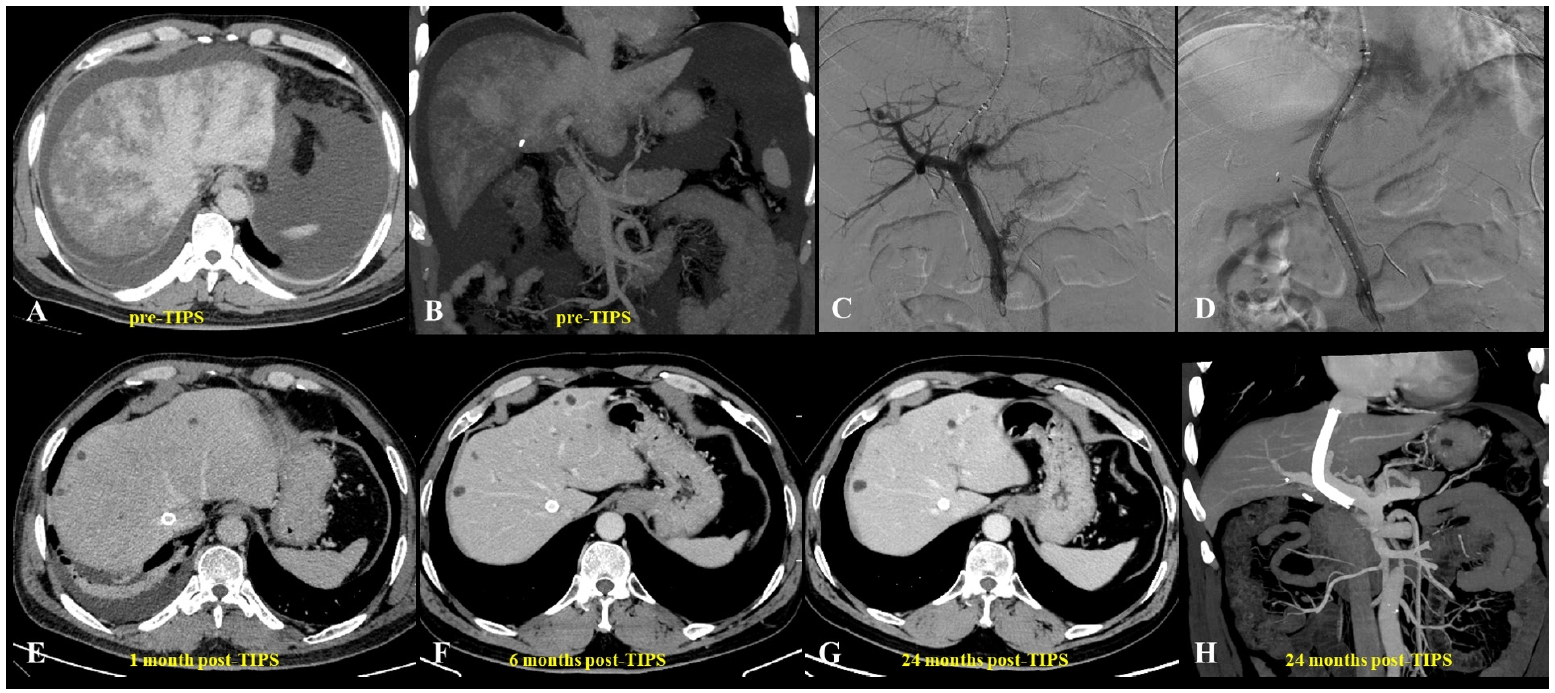
Fig. 1.A 62-year-old man diagnosed with hepatic sinusoidal obstruction syndrome 4 months after ingestion of Gynura segetum. (A) Abdominal computed tomography (CT) during the hepatic venous phase demonstrated patchy parenchymal enhancement with characteristic clover-like enhancement around the hepatic veins. Significant ascites and bilateral pleural effusions were also observed. (B) Coronal CT revealed patchy liver enhancement and narrowing (thinning) of the portal vein, accompanied by abundant ascites. (C, D) Portography before and after transjugular intrahepatic portosystemic shunt (TIPS) creation. A guidewire, introduced via the right hepatic artery, was used to provide real-time guidance during the procedure. (E–H) Post-TIPS abdominal CT showed well-defined opacification of hepatic veins with substantial resolution of both ascites and pleural effusions. Long-term follow-up CT, extending up to 24 months, confirmed homogeneous enhancement of the liver parenchyma and sustained patency of the TIPS shunt.
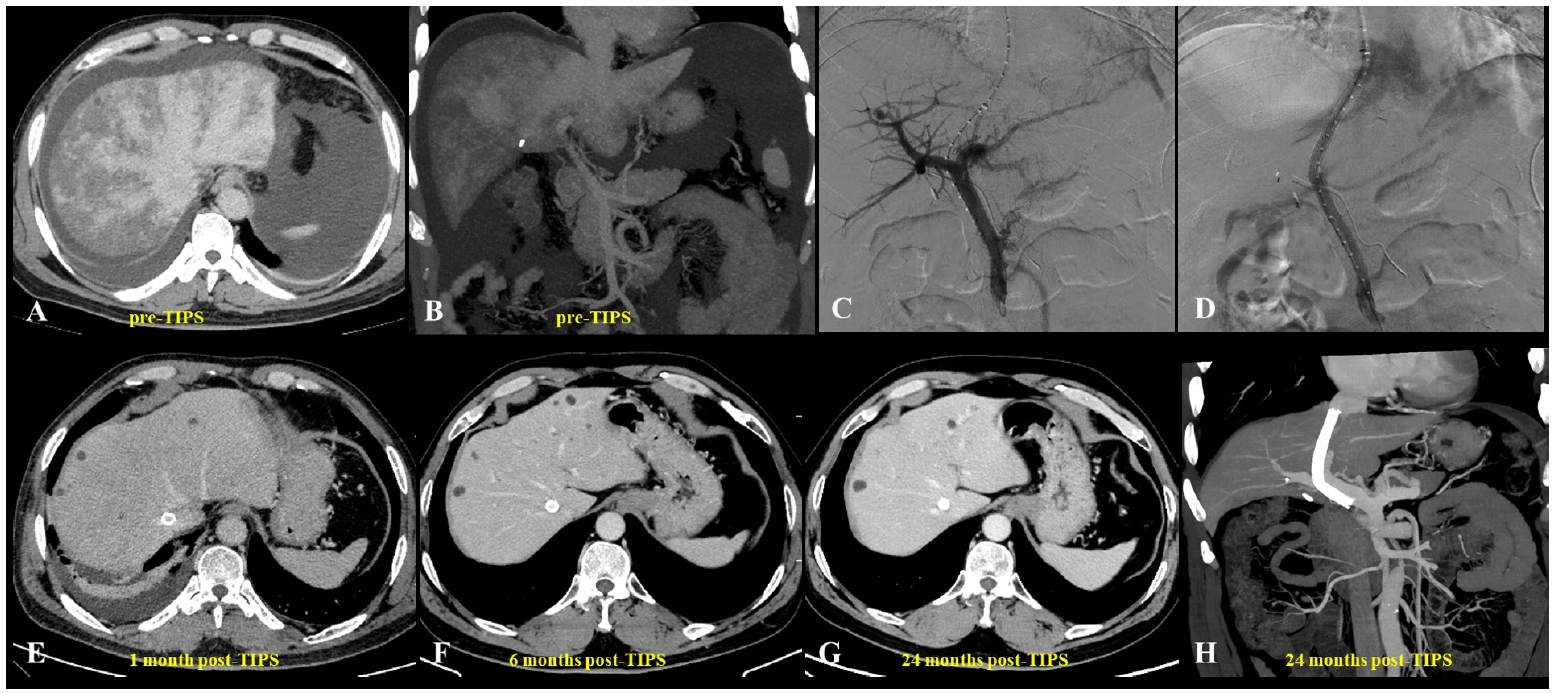
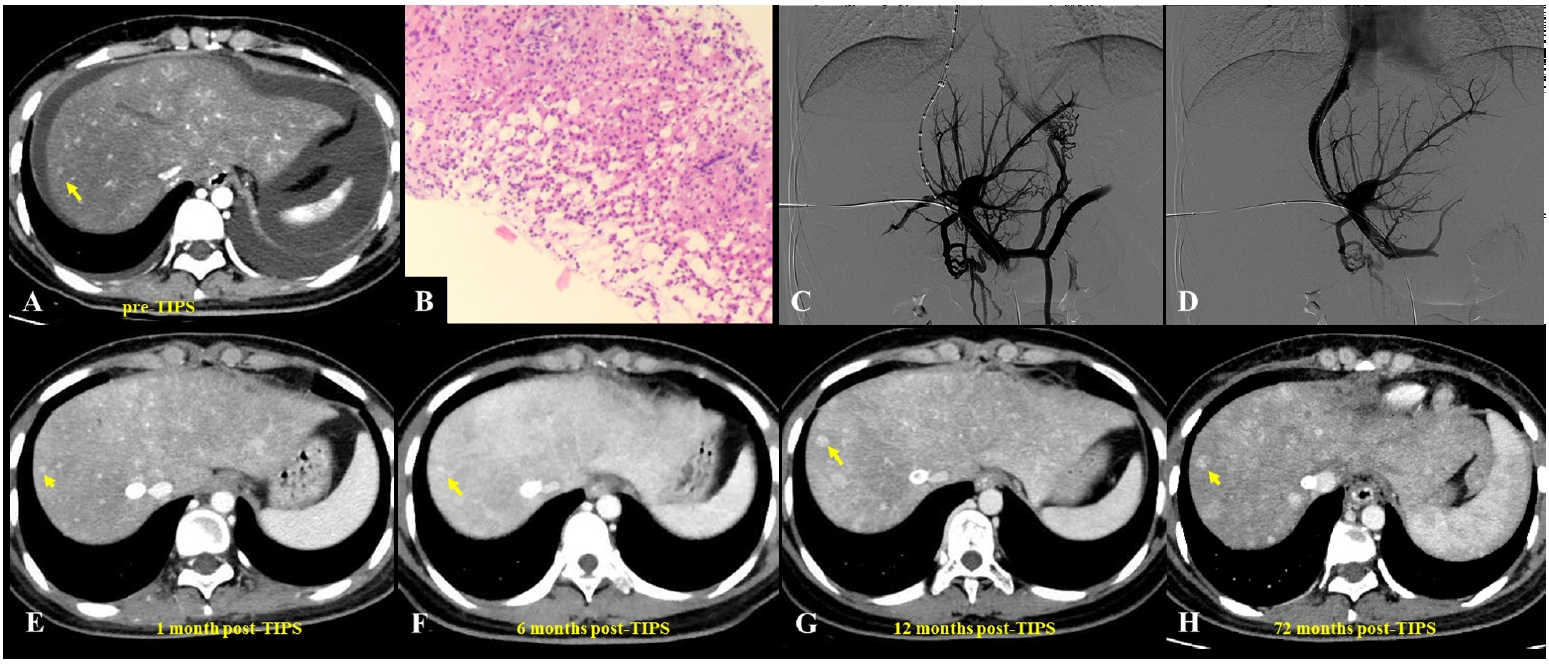
Fig. 2.A 23-year-old woman presented with pyrrolizidine alkaloid–induced hepatic sinusoidal obstruction syndrome following ingestion of oral weight-loss medications. (A) Pre–transjugular intrahepatic portosystemic shunt (TIPS) computed tomography (CT) (portal venous phase) demonstrated heterogeneous, patchy hepatic enhancement with multiple regenerative nodules (yellow arrow), accompanied by massive ascites and bilateral pleural effusions. (B) Histopathological analysis from a transjugular liver biopsy revealed focal hepatic atrophy, marked sinusoidal dilatation, and hepatocellular cholestasis, consistent with hepatic venous outflow obstruction. Immunohistochemical staining was negative for hepatitis B surface antigen, hepatitis B core antigen, diastase-periodic acid-Schiff, and periodic acid-Schiff, while showing positivity for cytokeratin 7 (biliary epithelium), reticulin, and Masson’s trichrome (indicating underlying fibrosis) (H&E, ×400). (C) Due to the diminished caliber (slenderness) of the portal vein, percutaneous transhepatic balloon-assisted TIPS placement was performed. Initial portal venography confirmed narrowing of the intrahepatic portal branches, stagnant portal flow, and the presence of prominent esophagogastric varices. (D) Post-TIPS venography demonstrated a widely patent shunt with unobstructed portal venous return and successful decompression of the varices. (E-H) Follow-up contrast-enhanced CT scans (venous phase) obtained at 1 month (E), 6 months (F), 12 months (G), and up to 72 months (H) post-TIPS demonstrated persistent heterogeneous enhancement and intrahepatic nodules (yellow arrows).
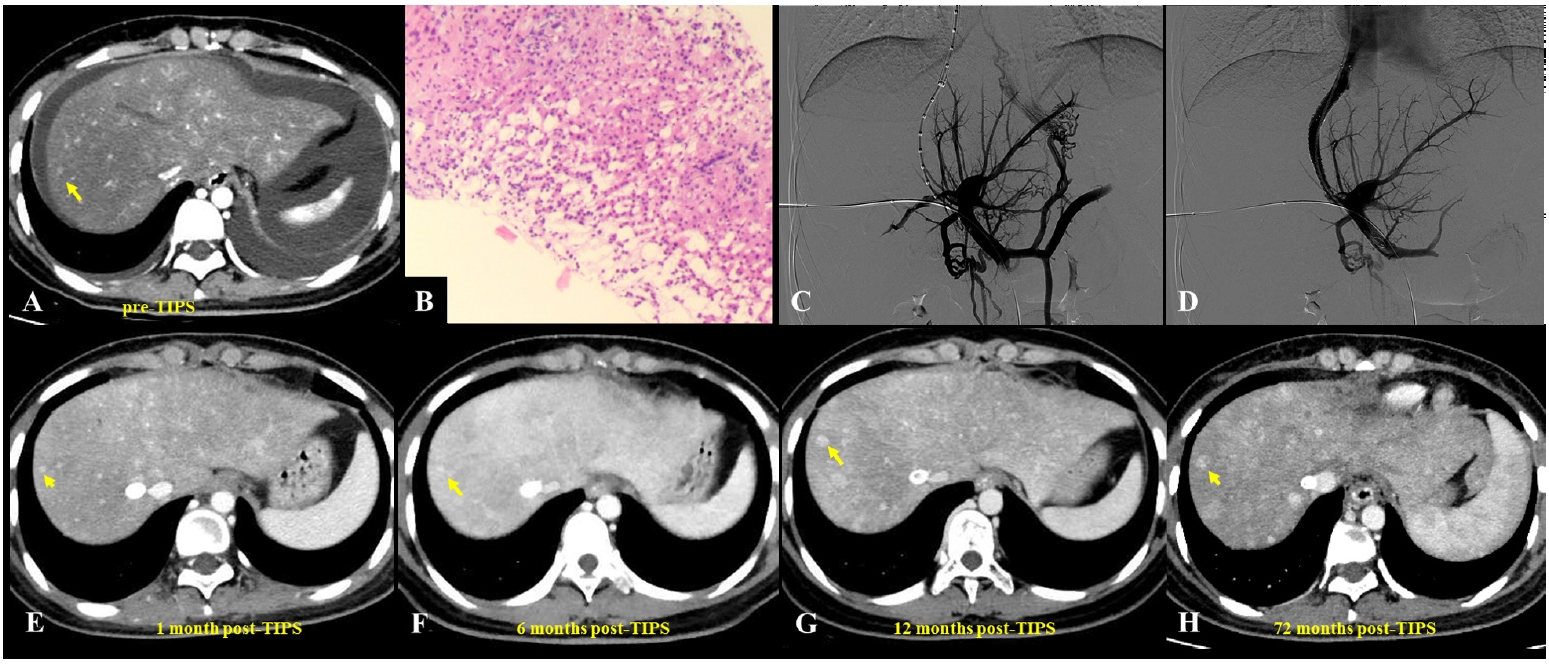
Table 1.Common plants causing PA-HSOS (Asia region)
Table 1.
|
Plant family |
Common genera/species |
Notes |
|
Asteraceae (Compositae) |
Gynura japonica (Tusanqi) |
Leading cause of PA-HSOS in China; roots/herbs used in traditional medicine |
|
Gynura segetum
|
Widely used herbal plant containing PAs; linked to HSOS in China |
|
Senecio spp. |
PAs (e.g., retrorsine) damage sinusoidal endothelial cells |
|
Tussilago farfara (coltsfoot) |
Traditional herb with PAs; potential HSOS risk |
|
Boraginaceae |
Heliotropium spp. |
Plants containing hepatotoxic PAs; implicated in HSOS cases |
|
Symphytum spp. (comfrey) |
Traditional medicinal herb; PAs cause HSOS and chronic liver damage |
|
Echium spp. |
PA-containing plants associated with liver injury |
|
Fabaceae (Leguminosae) |
Crotalaria spp. |
Plants containing monocrotaline, a PA that induces sinusoidal obstruction in animal models |
Table 2.Key features for differential diagnosis of PA-HSOS, Budd-Chiari syndrome, and typical cirrhotic decompensation
Table 2.
|
Feature |
PA-HSOS |
Budd-Chiari syndrome |
Typical cirrhotic decompensation |
|
Etiology |
Exposure to PAs |
Thrombosis of large hepatic veins or inferior vena cava |
Chronic liver disease (e.g., viral hepatitis, alcohol) |
|
Onset |
Acute/subacute |
Acute or subacute |
Chronic, with acute exacerbations |
|
Portal hypertension |
Yes, post-sinusoidal portal hypertension due to hepatic outflow obstruction |
Yes, due to obstruction of major veins |
Yes, due to cirrhosis |
|
Imaging findings |
Patchy parenchymal enhancement on CT/MRI |
Hepatic vein occlusion, collateral circulation |
Nodular liver surface, ascites, and splenomegaly on imaging |
|
Ascites |
Common, often refractory to diuretics |
Common, often with significant fluid accumulation |
Common, usually associated with other signs of cirrhosis |
|
Clinical presentation |
Ascites, hepatomegaly, jaundice |
Acute liver failure, abdominal pain, ascites |
Varices, jaundice, hepatic encephalopathy |
|
Histopathology |
Sinusoidal dilatation, necrosis, fibrosis |
Thrombosis-related changes in the liver |
Fibrosis, cirrhosis, regenerative nodules |
|
Coagulation status |
Often coagulopathy present |
Prothrombotic states common |
Coagulopathy may be present due to liver dysfunction |
|
Risk factors |
Herbal medicine use, exposure to specific plants |
Myeloproliferative disorders, pregnancy, oral contraceptives |
Alcohol use, viral hepatitis, metabolic disorders |
|
Management |
Supportive care, TIPS for refractory cases |
Anticoagulation, TIPS for severe cases |
Management of underlying liver disease, TIPS for refractory ascites |
Table 3.Summary of major studies on TIPS for PA-HSOS in Asia
Table 3.
|
Country |
Author (Year) |
Sample size |
Type of study |
Aim of study |
Outcomes |
|
China |
Dai et al. (2025) [52] |
30 Patients |
Retrospective analysis |
Evaluated safety/efficacy of TIPS |
The PPG significantly decreased post-TIPS, with ascites remission occurring in 52 days (median) and liver CT showing recovery in 196.5 days. Disease severity and ALBI grade were key recovery time predictors. |
|
China |
Huang et al. (2023) [50] |
20 Patients (TIPS) and 41 patients (AT) |
Comparative study |
Compared mid-long-term outcomes of TIPS vs. AT as initial treatment |
TIPS demonstrated superior mid- to long-term outcomes compared to anticoagulation therapy, particularly for patients with severe or very severe PA-induced HSOS. |
|
China |
Wu et al. (2021) [53] |
4 in 10 PA-HSOS patients received TIPS |
Retrospective analysis |
Evaluated TIPS timing using TBIL as a measure |
TIPS timing based on TBIL levels affected efficacy. |
|
China |
Huang et al. (2023) [57] |
12 Patients (TIPS) and 10 patients (conservative treatment) |
Retrospective cohort |
Investigated efficacy and safety of TIPS in PA-HSOS |
TIPS may be a safe and effective therapeutic strategy for PA-HSOS patients who do not respond to conservative treatment. |
|
China |
Zhou et al. (2020) [56] |
37 Patients (TIPS) and 17 patients (conservative treatment) |
Retrospective analysis |
Evaluated TIPS as treatment for PA-HSOS |
Better outcomes may be achieved with TIPS compared with conventional symptomatic treatment in patients with PA-HSOS. |
|
China |
Wang et al. (2024) [54] |
164 Patients (69 in TIPS group vs. 95 in supportive group) |
Retrospective analysis |
Compared clinical outcomes and liver histology between TIPS and supportive treatment |
In comparison with supportive treatment, TIPS treatment improved clinical outcomes and liver histology. |
|
China |
Xiao et al. (2021) [55] |
116 Patients |
Retrospective analysis |
Evaluated predictors of poor outcomes in PA-HSOS patients receiving TIPS |
Prolonged prothrombin time at baseline and increased serum TBIL levels 5 days after TIPS were independent risk factors for predicting death after TIPS treatment in PA-HSOS patients. |
References
- 1. Yu Z, Li W, Tian C, Cao Y, Zhang C. Drug-induced hepatic sinusoidal obstruction syndrome: current advances and future perspectives. Arch Toxicol. 2025;99:835-850. https://doi.org/10.1007/s00204-024-03950-9
- 2. Shukla A, Rockey DC, Kamath PS, Kleiner DE, Singh A, Vaidya A, et al. Non-cirrhotic portal fibrosis/idiopathic portal hypertension: APASL recommendations for diagnosis and management. Hepatol Int. 2024;18:1684-1711. https://doi.org/10.1007/s12072-024-10739-6
- 3. Venkatesh SK, Harper KC, Borhani AA, Furlan A, Thompson SM, Chen EZ, et al. Hepatic sinusoidal disorders. Radiographics. 2024;44:e240006. https://doi.org/10.1148/rg.240006
- 4. Kaca do Carmo LH, Freitas MT, Colla RB, Verdan S, Bando TY, Andrade M, et al. Transient and shear-wave elastography in the detection of hepatic sinusoidal obstruction syndrome in patients undergoing hematopoietic stem cell transplantation: a systematic review and meta-analysis. Transplant Cell Ther. 2025;31:1043. https://doi.org/10.1016/j.jtct.2025.09.004
- 5. Larue M, Malard F, Alaskar AS, Aljurf M, Arat M, Balsat M, et al. Management of liver sinusoidal obstruction syndrome/veno-occlusive disease in adults: a 2025 perspective from an international expert group. Bone Marrow Transplant. 2025;60:1002-1008. https://doi.org/10.1038/s41409-025-02598-y
- 6. Zong J, Shen T, Mei Y, Liu F, Li QX, Jiao W. Absorption, distribution, and metabolism of pyrrolizidine alkaloids in tea plants: insights from hydroponic exposure and molecular simulations. Food Chem. 2025;493:145887. https://doi.org/10.1016/j.foodchem.2025.145887
- 7. Yang XQ, Ye J, Li X, Li Q, Song YH. Pyrrolizidine alkaloids-induced hepatic sinusoidal obstruction syndrome: pathogenesis, clinical manifestations, diagnosis, treatment, and outcomes. World J Gastroenterol. 2019;25:3753-3763. https://doi.org/10.3748/wjg.v25.i28.3753
- 8. Liu Z, Liang S, Wei X, Du X, Zhang J. Defibrotide improved the outcome of monocrotaline induced rat hepatic sinusoidal obstruction syndrome. BMC Gastroenterol. 2022;22:525. https://doi.org/10.1186/s12876-022-02523-3
- 9. Xia C, Cen Y, Yao S, Xu S, Lou G, Liu Y, et al. Monocrotaline induces liver injury via TRX1-ASK1-JNK axis-mediated mitochondrial damage in hepatocytes. Int Immunopharmacol. 2026;172:116121. https://doi.org/10.1016/j.intimp.2025.116121
- 10. Yang P, Zhang F, Wang F, He Y, Wang Y, Qian L. Transjugular intrahepatic portosystemic shunt for hepatic sinusoidal obstruction syndrome with primary biliary cholangitis and alcoholic liver disease: a case report. Front Med (Lausanne). 2025;12:1696892. https://doi.org/10.3389/fmed.2025.1696892
- 11. Li R, Li L, Cai Z, Chen J, Zhang H, Zhao S, et al. Histological evaluation of pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome: correlation with Drum Tower Severity Scoring. Dig Liver Dis. 2024;56:1220-1228. https://doi.org/10.1016/j.dld.2023.12.008
- 12. Zong T, Li M, Hu Z, Jin L, Liu Y, Duan Y, et al. Traditional uses, phytochemistry, pharmacology, and toxicology of Belamcanda chinensis: a review. Plants (Basel). 2025;14:3688. https://doi.org/10.3390/plants14233688
- 13. Chahrour JA, Abdel Baki Z, El Badan D, Nasser G, Maresca M, Hijazi A. Herbal medicines in the management of diabetes mellitus: plants, bioactive compounds, and mechanisms of action. Biomolecules. 2025;15:1674. https://doi.org/10.3390/biom15121674
- 14. Bessaire T, Mujahid C, Mottier P, Du K, Savage A, Fu X, et al. Simultaneous and quantitative determination of pyrrolizidine and tropane alkaloids in food by LC-MS/MS, first action 2025.02. J AOAC Int. 2026;109:99-117. https://doi.org/10.1093/jaoacint/qsaf097
- 15. Zhuge Y, Liu Y, Xie W, Zou X, Xu J, Wang J, et al. Expert consensus on the clinical management of pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. J Gastroenterol Hepatol. 2019;34:634-642. https://doi.org/10.1111/jgh.14612
- 16. Jang S, Kim KH, Sun SH, Go HY, Lee EK, Jang BH, et al. Characteristics of herbal medicine users and adverse events experienced in South Korea: a survey study. Evid Based Complement Alternat Med. 2017;2017:4089019. https://doi.org/10.1155/2017/4089019
- 17. Ravi S, Bharadvaja N. Market analysis of medicinal plants in India. Curr Pharm Biotechnol. 2019;20:1172-1180. https://doi.org/10.2174/1389201020666190819154516
- 18. Huang CW, Hwang IH, Yun YH, Jang BH, Chen FP, Hwang SJ, et al. Population-based comparison of traditional medicine use in adult patients with allergic rhinitis between South Korea and Taiwan. J Chin Med Assoc. 2018;81:708-713. https://doi.org/10.1016/j.jcma.2017.12.008
- 19. Srirama R, Santhosh Kumar JU, Seethapathy GS, Newmaster SG, Ragupathy S, Ganeshaiah KN, et al. Species adulteration in the herbal trade: causes, consequences and mitigation. Drug Saf. 2017;40:651-661. https://doi.org/10.1007/s40264-017-0527-0
- 20. Unnikrishnan R, Dev SA, Jayaraj R. Pitfalls and promises of raw drug identification techniques in the ayurvedic industry: an overview. 3 Biotech. 2020;10:497. https://doi.org/10.1007/s13205-020-02482-0
- 21. Zhang S, Yan D, Cheng S, Jin J, Cui J, Liu C, et al. Gancao decoction ameliorated senecionine-induced hepatic sinusoidal obstruction syndrome in mice by inhibiting NET formation and senecionine bioactivation in liver. J Ethnopharmacol. 2026;362:121356. https://doi.org/10.1016/j.jep.2026.121356
- 22. Tan Y, Zheng S. Clinicopathological characteristics and diagnosis of hepatic sinusoidal obstruction syndrome caused by Tusanqi: case report and literature review. Open Med (Wars). 2023;18:20230737. https://doi.org/10.1515/med-2023-0737
- 23. Zhang C, Zhang Q, Wang P, Yin H, Liu Y, Zhang S, et al. Retrorsine-induced hepatotoxicity is mediated by inhibition of the EGFR/AKT/c-Jun axis and disruption of calcium homeostasis in primary hepatocytes. Toxicon. 2026;272:108998. https://doi.org/10.1016/j.toxicon.2026.108998
- 24. Yao J, Qin Y, Tu D, Huang Y, Liang L, Cai W, et al. Combined metabolome and transcriptome to analyze the regulatory network of key enzymes in the synthesis of senkirkine in Emilia sonchifolia. BMC Plant Biol. 2025;25:1006. https://doi.org/10.1186/s12870-025-07079-4
- 25. Gao B, Zhang J, Zhu L, Zhang Y. Radical total gastrectomy for gastric cancer complicated by hepatic sinusoidal obstruction syndrome: a case report. Front Med (Lausanne). 2025;12:1544400. https://doi.org/10.3389/fmed.2025.1544400
- 26. Zhu L, Zhang CY, Li DP, Chen HB, Ma J, Gao H, et al. Tu-San-Qi (Gynura japonica): the culprit behind pyrrolizidine alkaloid-induced liver injury in China. Acta Pharmacol Sin. 2021;42:1212-1222. https://doi.org/10.1038/s41401-020-00553-9
- 27. Wang X, Zhang W, Zhang M, Zhang F, Xiao J, Yin Q, et al. Development of a Drum Tower Severity Scoring (DTSS) system for pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. Hepatol Int. 2022;16:669-679. https://doi.org/10.1007/s12072-021-10293-5
- 28. Yang X, Wang H, Ni HM, Xiong A, Wang Z, Sesaki H, et al. Inhibition of Drp1 protects against senecionine-induced mitochondria-mediated apoptosis in primary hepatocytes and in mice. Redox Biol. 2017;12:264-273. https://doi.org/10.1016/j.redox.2017.02.020
- 29. Terkelsen MK, Bendixen SM, Hansen D, Scott EA, Moeller AF, Nielsen R, et al. Transcriptional dynamics of hepatic sinusoid-associated cells after liver injury. Hepatology. 2020;72:2119-2133. https://doi.org/10.1002/hep.31215
- 30. Gibert-Ramos A, Andres-Rozas M, Pasto R, Alfaro-Retamero P, Guixe-Muntet S, Gracia-Sancho J. Sinusoidal communication in chronic liver disease. Clin Mol Hepatol. 2025;31:32-55. https://doi.org/10.3350/cmh.2024.0734
- 31. Xu J, Xue Q, Xiong A, Chen Y, Wang X, Yan X, et al. Chlorogenic acid attenuates pyrrolizidine alkaloid-induced liver injury through modulation of the SIRT1/FXR signaling pathway. Chin Med. 2025;20:34. https://doi.org/10.1186/s13020-025-01077-2
- 32. Huang Z, Wu Z, Gu X, Ji L. Diagnosis, toxicological mechanism, and detoxification for hepatotoxicity induced by pyrrolizidine alkaloids from herbal medicines or other plants. Crit Rev Toxicol. 2024;54:123-133. https://doi.org/10.1080/10408444.2024.2310597
- 33. Shang H, Huang C, Xiao Z, Yang P, Zhang S, Hou X, et al. Gut microbiota-derived tryptophan metabolites alleviate liver injury via AhR/Nrf2 activation in pyrrolizidine alkaloids-induced sinusoidal obstruction syndrome. Cell Biosci. 2023;13:127. https://doi.org/10.1186/s13578-023-01078-4
- 34. Zhao S, Zhang H, Zhu H, Zhao T, Tu J, Yin X, et al. Gut microbiota promotes macrophage M1 polarization in hepatic sinusoidal obstruction syndrome via regulating intestinal barrier function mediated by butyrate. Gut Microbes. 2024;16:2377567. https://doi.org/10.1080/19490976.2024.2377567
- 35. Chen X, Ma J, He Y, Xue J, Song Z, Xu Q, et al. Characterization of liver injury induced by a pyrrolizidine alkaloid in rats. Phytomedicine. 2021;89:153595. https://doi.org/10.1016/j.phymed.2021.153595
- 36. Xiong A, Shao Y, Fang L, Yang X, Zhang S, Zheng J, et al. Comparative analysis of toxic components in different medicinal parts of Gynura japonica and its toxicity assessment on mice. Phytomedicine. 2019;54:77-88. https://doi.org/10.1016/j.phymed.2018.06.015
- 37. Du X, Liu Z, Yu H, Wang Y, Zou Z, Wei H, et al. Prognostic risk factors for patients with hepatic sinusoidal obstruction syndrome caused by pyrrolizidine alkaloids. Medicine (Baltimore). 2023;102:e34698. https://doi.org/10.1097/MD.0000000000034698
- 38. Shang H, Bai T, Zhu S, Yang X, Liu C, Xu D, et al. Prognostic factors for pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome: a multicenter study in China. Ann Transl Med. 2021;9:11. https://doi.org/10.21037/atm-20-731
- 39. Peng C, Zhang X, Zhang F, Liu L, Shao Y, Xiang X, et al. Clinical efficacy and safety of anticoagulation therapy for Pyrrolizidine alkaloids-induced hepatic sinusoidal obstruction syndrome: a retrospective multicenter cohort study. Eur J Gastroenterol Hepatol. 2020;32:1168-1178. https://doi.org/10.1097/MEG.0000000000001630
- 40. Kurimoto M, Chang TC, Nishiyama Y, Suzuki T, Dohmae N, Tanaka K, et al. Anticancer approach inspired by the hepatotoxic mechanism of pyrrolizidine alkaloids with glycosylated artificial metalloenzymes. Angew Chem Int Ed Engl. 2022;61:e202205541. https://doi.org/10.1002/anie.202205541
- 41. He Y, Zhu L, Ma J, Lin G. Metabolism-mediated cytotoxicity and genotoxicity of pyrrolizidine alkaloids. Arch Toxicol. 2021;95:1917-1942. https://doi.org/10.1007/s00204-021-03060-w
- 42. Widjaja-van den Ende F, van Boekel M, Davis C, Wesseling S, Rietjens I. Quantifying the effect of human interindividual kinetic differences on the relative potency value for riddelliine N-oxide at low dose levels by a new approach methodology. Regul Toxicol Pharmacol. 2025;156:105767. https://doi.org/10.1016/j.yrtph.2024.105767
- 43. Zhang LL, Zhang F, Wang K, Song YH, Zhang Y, Zhou ZY, et al. Validation of Drum Tower Severity Scoring (DTSS) system for pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. J Dig Dis. 2025;26:150-157. https://doi.org/10.1111/1751-2980.13347
- 44. Zhou H, Wang YX, Lou HY, Xu XJ, Zhang MM. Hepatic sinusoidal obstruction syndrome caused by herbal medicine: CT and MRI features. Korean J Radiol. 2014;15:218-225. https://doi.org/10.3348/kjr.2014.15.2.218
- 45. Liu F, Rong X, Guo H, Xu D, Liu C, Meng L, et al. Clinical characteristics, CT signs, and pathological findings of pyrrolizidine alkaloids-induced sinusoidal obstructive syndrome: a retrospective study. BMC Gastroenterol. 2020;20:30. https://doi.org/10.1186/s12876-020-1180-0
- 46. Guo T, Li X, Yang X, Kong X, Liu H, Bai T, et al. Gadoxetic acid-enhanced hepatobiliary-phase magnetic resonance imaging for pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome and association with liver function. Sci Rep. 2019;9:1231. https://doi.org/10.1038/s41598-018-37775-1
- 47. Tong Y, Zhang M, Qi Z, Wu W, Chen J, He F, et al. Hepatic venous occlusion type of Budd-Chiari syndrome versus pyrrolizidine alkaloid-induced hepatic sinusoidal obstructive syndrome: a multi-center retrospective study. J Pers Med. 2023;13:603. https://doi.org/10.3390/jpm13040603
- 48. European Association for the Study of the Liver. EASL clinical practice guidelines on vascular diseases of the liver. J Hepatol. 2026;84:399-456. https://doi.org/10.1016/j.jhep.2025.08.001
- 49. Jiang M, Wang L, Du X, Hao M, Gao P. Low molecular weight heparin in the treatment of pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome: five case reports. J Int Med Res. 2020;48:300060520961916. https://doi.org/10.1177/0300060520961916
- 50. Huang Q, Zhang Q, Xu H, Zu M, Xiao J, Shen B. Mid- to long-term outcomes of initial transjugular intrahepatic portosystemic shunt versus anticoagulation for pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. Eur J Gastroenterol Hepatol. 2023;35:445-452. https://doi.org/10.1097/MEG.0000000000002509
- 51. Kernan NA, Grupp S, Smith AR, Arai S, Triplett B, Antin JH, et al. Final results from a defibrotide treatment-IND study for patients with hepatic veno-occlusive disease/sinusoidal obstruction syndrome. Br J Haematol. 2018;181:816-827. https://doi.org/10.1111/bjh.15267
- 52. Dai J, Yang X, Hu X, Deng Y, Lu J, Wang J, et al. Albumin-bilirubin and severity as key factors of recovery in patients with PA-HSOS undergoing transjugular intrahepatic portosystemic shunt. Ann Ital Chir. 2025;96:1018-1027. https://doi.org/10.62713/aic.4033
- 53. Wu F, Yu J, Gan H, Zhang H, Tian D, Zheng D. Timing and efficacy of transjugular intrahepatic portosystemic shunt in patients with pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. Sci Rep. 2021;11:21743. https://doi.org/10.1038/s41598-021-01201-w
- 54. Wang C, Wang Y, Zhao J, Yang C, Zhu X, Niu H, et al. Transjugular intrahepatic portosystemic shunt for the treatment of hepatic sinusoidal obstruction syndrome caused by pyrrolizidine alkaloids: a multicenter retrospective study. Heliyon. 2024;10:e23455. https://doi.org/10.1016/j.heliyon.2023.e23455
- 55. Xiao J, Tu J, Zhang H, Zhang F, Zhang W, Xu H, et al. Risk factors of poor prognosis in patients with pyrrolidine alkaloid-induced hepatic sinusoidal obstruction syndrome after transjugular intrahepatic portosystemic shunt. Hepatol Int. 2021;15:720-729. https://doi.org/10.1007/s12072-020-10126-x
- 56. Zhou CZ, Wang RF, Lv WF, Fu YQ, Cheng DL, Zhu YJ, et al. Transjugular intrahepatic portosystemic shunt for pyrrolizidine alkaloid-related hepatic sinusoidal obstruction syndrome. World J Gastroenterol. 2020;26:3472-3483. https://doi.org/10.3748/wjg.v26.i24.3472
- 57. Huang T, Zhang X, Yan K, Lou D, He Y, Dai S, et al. Transjugular intrahepatic portosystemic shunt for pyrrolidine alkaloids-induced hepatic sinusoidal obstruction syndrome: a retrospective cohort study. Eur J Gastroenterol Hepatol. 2023;35:1004-1011. https://doi.org/10.1097/MEG.0000000000002591
- 58. Cen P, Ding J, Jin J. Hepatic sinusoidal obstruction syndrome caused by the ingestion of Gynura segetum in a patient with alcoholic cirrhosis: a case report. J Int Med Res. 2021;49:300060520980649. https://doi.org/10.1177/0300060520980649
- 59. Li S, Li Y, Zhou C, Li H, Chen C, Peng C, et al. Transjugular intrahepatic portosystemic shunt benefits for hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum: a propensity score-matched analysis. Cardiovasc Intervent Radiol. 2023;46:931-942. https://doi.org/10.1007/s00270-023-03451-9
- 60. Cheng Y, Gu L, Yin X, Wang X, Xiao J, Wang Y, et al. Agreement between wedged hepatic venous pressure and portal pressure in hepatic sinusoidal obstruction syndrome. J Pers Med. 2022;13:4. https://doi.org/10.3390/jpm13010004
- 61. Zhang L, Li Q, Makamure J, Zhao D, Liu Z, Zheng C, et al. Transjugular intrahepatic portosystemic shunt for hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. BMC Gastroenterol. 2021;21:26. https://doi.org/10.1186/s12876-021-01599-7
- 62. Luo S, Chu J, Huang H, Yao K. Direct intrahepatic portocaval shunt for sinusoidal obstruction syndrome associated with hepatotoxicity of pyrrolizidine alkaloids. Biomed Res Int. 2018;2018:9804582. https://doi.org/10.1155/2018/9804582
- 63. Roccarina D, Saltini D, Senzolo M, Nardelli S, Rosi M, Adotti V, et al. Shunt magnitude is a key determinant of overt hepatic encephalopathy in patients undergoing TIPS. JHEP Rep. 2026;8:101676. https://doi.org/10.1016/j.jhepr.2025.101676
- 64. Jiang X, Ma X, Tian S, Peng L. Efficacy and safety of transjugular intrahepatic portosystemic shunt in hepatic sinusoidal obstruction syndrome: systematic review and meta-analysis. Front Med (Lausanne). 2025;12:1625825. https://doi.org/10.3389/fmed.2025.1625825
- 65. Xiang Y, Tie J, Wang G, Zhuge Y, Wu H, Zhu X, et al. Post-TIPS overt hepatic encephalopathy increases long-term but not short-term mortality in cirrhotic patients with variceal bleeding: a large-scale, multicenter real-world study. Aliment Pharmacol Ther. 2025;61:1183-1196. https://doi.org/10.1111/apt.18509
- 66. Will V, Rodrigues SG, Stirnimann G, Gottardi A, Bosch J, Berzigotti A. Transjugular intrahepatic portosystemic shunt and alfapump(R) system for refractory ascites in liver cirrhosis: outcomes and complications. United European Gastroenterol J. 2020;8:961-969. https://doi.org/10.1177/2050640620938525
- 67. Zhang W, Liu L, Zhang M, Zhang F, Peng C, Zhang B, et al. Validation of the Nanjing criteria for diagnosing pyrrolizidine alkaloids-induced hepatic sinusoidal obstruction syndrome. J Clin Transl Hepatol. 2021;9:345-352. https://doi.org/10.14218/JCTH.2020.00124
- 68. Yang L, Ju H, Chen Z, Cheng S, Liu Y, Wang X. PU.1 aggravates hepatic sinusoidal obstruction syndrome by upregulating PTBP1 and activating the Wnt/beta-catenin pathway. Histol Histopathol. 2026;41:305-318. https://doi.org/10.14670/HH-18-949
- 69. He Y, Ma J, Fan X, Ding L, Ding X, Zhang QY, et al. The key role of gut-liver axis in pyrrolizidine alkaloid-induced hepatotoxicity and enterotoxicity. Acta Pharm Sin B. 2021;11:3820-3835. https://doi.org/10.1016/j.apsb.2021.07.013
- 70. Gressens SB, Cazals-Hatem D, Lloyd V, Plessier A, Payance A, Lebrec D, et al. Hepatic venous pressure gradient in sinusoidal obstruction syndrome: diagnostic value and link with histological lesions. JHEP Rep. 2022;4:100558. https://doi.org/10.1016/j.jhepr.2022.100558
- 71. Sachse B, Hessel-Pras S, Schafer B. Genotoxic carcinogenicity of pyrrolizidine alkaloids: relevance of potency factors for the risk assessment. Arch Toxicol. 2026;100:95-108. https://doi.org/10.1007/s00204-025-04182-1